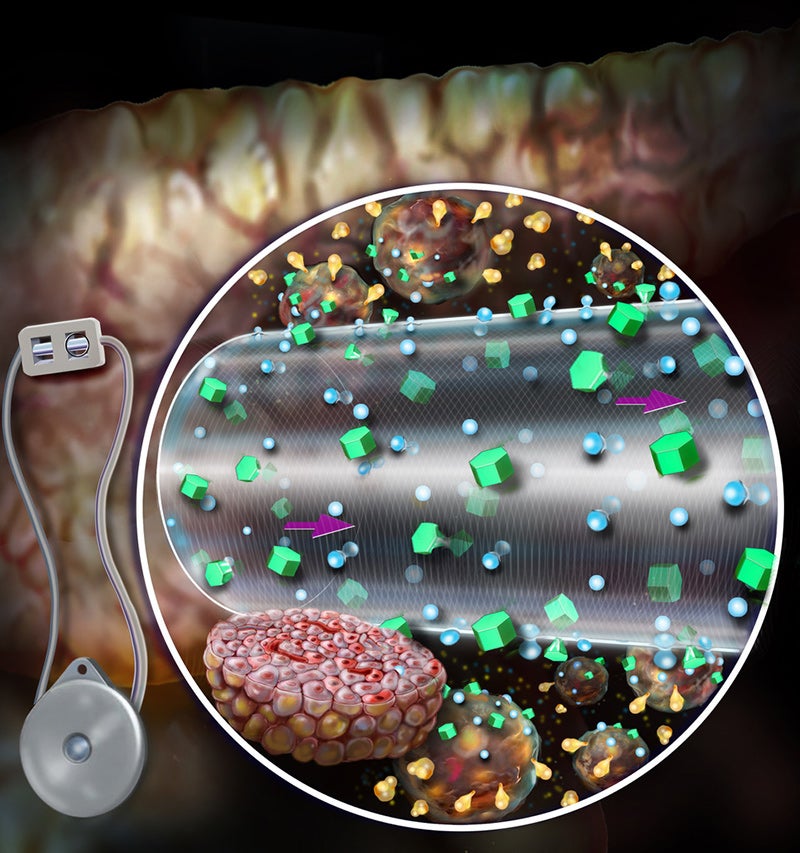
Pancryos has entered a worldwide exclusive licence agreement with Brigham and Women’s Hospital for the convection-enhanced macroencapsulation device (ceMED).
A team of researchers from Brigham and Women’s Hospital, Harvard Stem Cell Institute (HSCI), Harvard University and the University of Massachusetts Medical School developed ceMED.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
This device will act as the delivery mechanism for the company’s stem cell-derived, natural insulin production therapy, PanINSULA, for the treatment of type 1 diabetes.
It is intended to enhance cell loading capacity while boosting the survival of the cell, glucose sensitivity and timely secretion of insulin.
The device was shown to boost survival and insulin secretion of cells in preclinical models and started to reduce blood glucose levels as early as two days after transplantation.
When compared to current diffusion-based devices, ceMED offers substantial advantages, including better cell survival, decreased fibrous encapsulation, and faster on and off rates for insulin secretion.
According to initial findings obtained with the ceMED device, its distinctive design may be able to overcome the mass transport limitations of standard macroencapsulation devices and could also facilitate the delivery of greater doses to cells without affecting their viability or function.
Pancryos co-founder Henrik Semb said: “Our goal is to offer all patients with type 1 diabetes a safe and sustainable source of enriched immune-protected functional stem cell-derived beta cells.
“This exclusive licence puts Pancryos in a unique position to deliver our high-quality beta cells with the innovative ceMED device that will protect the cells from immune attack.
“This is a very exciting development for the treatment of Type 1 Diabetes and the millions who suffer without a cure.”
Impacting more than 50 million people globally, type 1 diabetes has no known treatment.




