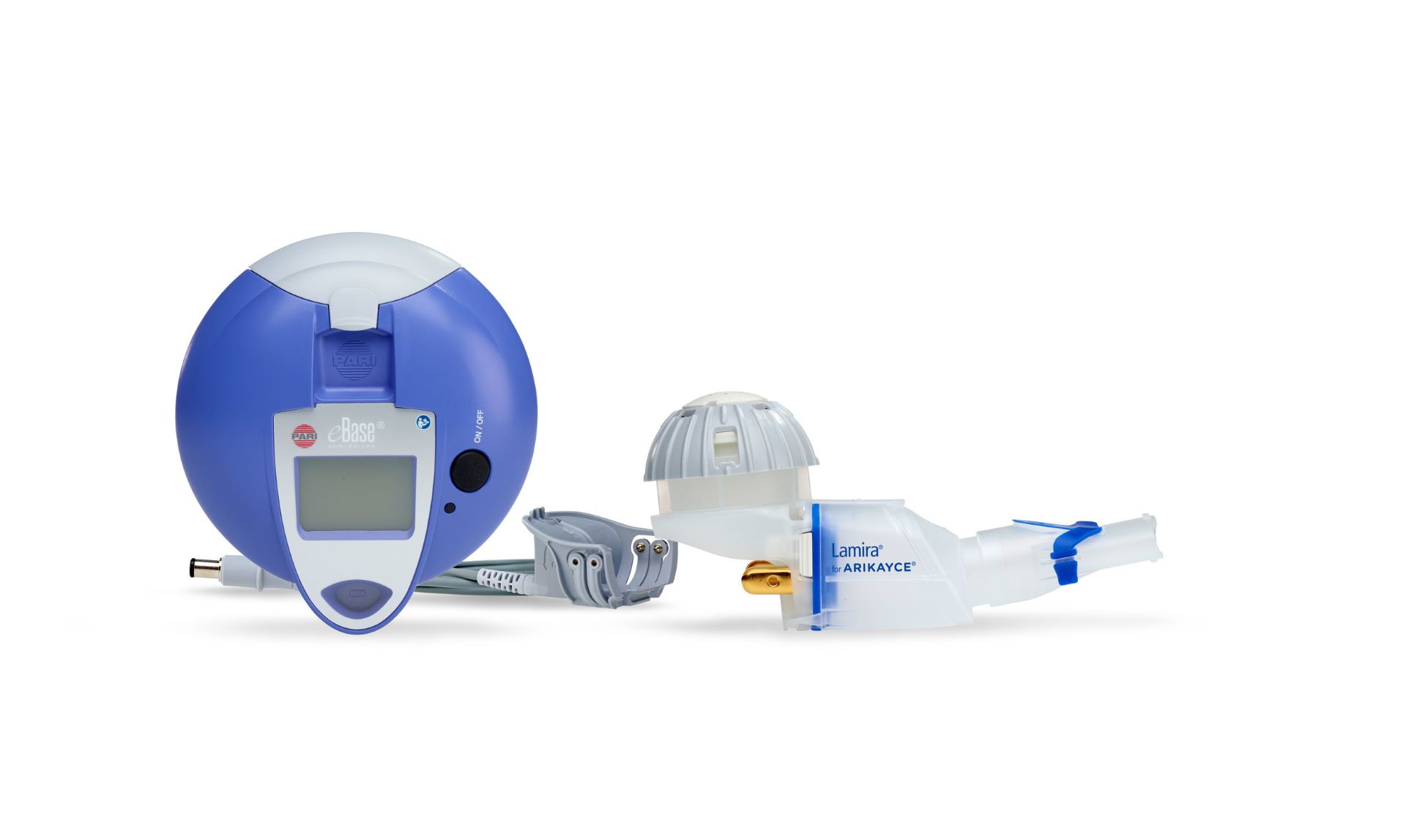
PARI Pharma has received authorisation for the Lamira Nebulizer System for delivery of Insmed’s drug product, ARIKAYCE (amikacin liposome inhalation suspension), in Japan.
The Lamira Nebulizer System has been specially optimised for administering ARIKAYCE.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
The drug-specific eFlow Technology nebuliser system comprises a customised medication reservoir that can hold a full 8.4ml dose, a tailored aerosol head for the aerosolisation of ARIKAYCE and the valved aerosol chamber.
PARI Pharma president Dr Martin Knoch said: “Lamira enables us the ability to offer an eFlow Technology nebuliser to patients beyond Europe and North America.
“We are delighted that appropriate patients in Japan suffering from Nontuberculous mycobacterial (NTM) lung disease now have access to a new treatment option.”
Earlier this week, Japan’s Ministry of Health, Labour and Welfare (MHLW) granted approval for ARIKAYCE following approvals in the US and Europe.
A novel, inhaled, once-daily formulation of amikacin, ARIKAYCE is claimed to be the first inhaled liposomal dispersion approved by the MHLW.
It is also claimed to be the only therapy in Japan for treating patients with NTM lung disease caused by Mycobacterium avium complex (MAC) who have not responded to a previous multidrug regimen treatment.
Insmed Japan general manager Yuji Orihara said: “We are pleased that our valuable partnership with PARI has enabled us to bring to market the first approved drug, ARIKAYCE, to be used with the LAMIRA nebuliser device specifically for the treatment of patients in Japan with refractory MAC lung disease.”
eFlow Technology is an aerosol delivery platform that aids in the effective nebulisation of liquid medications through a vibrating, perforated membrane.





