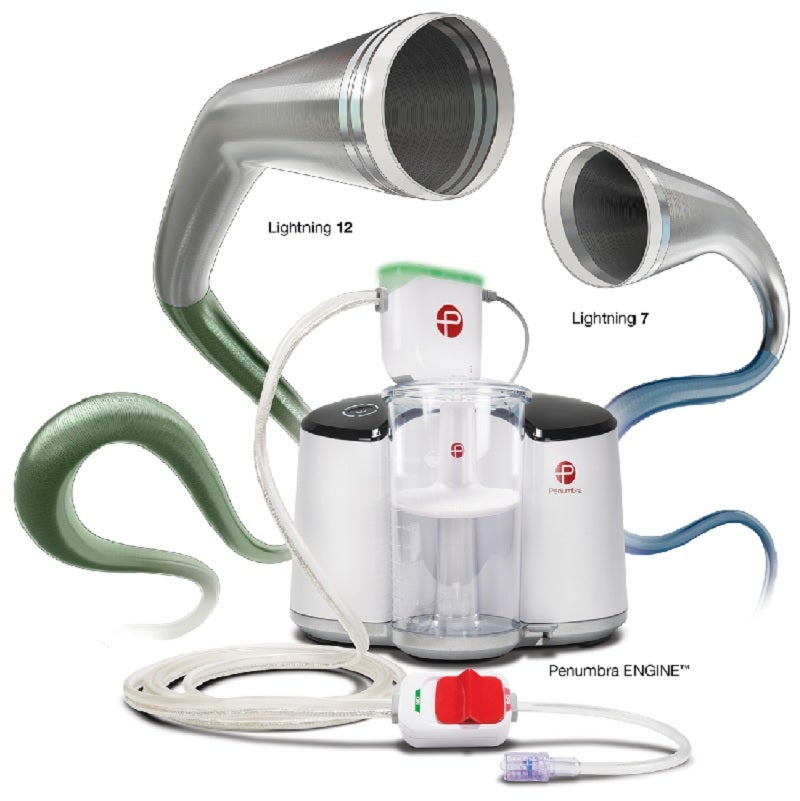
Healthcare company Penumbra has obtained CE Mark for its Indigo Aspiration System with Lightning 7 and Lightning 12 mechanical thrombectomy technologies.
Part of the Indigo system, the two technologies are designed for the removal of single session blood clot in peripheral arterial and venous systems, including the pulmonary embolisms treatment.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
They are powered by the Penumbra ENGINE and combine the new Indigo System CAT 7 and CAT 12 aspiration catheters with Lightning intelligent aspiration, a computer-aided clot detection technology.
With automatic valve control, Lightning intelligent aspiration has the ability to distinguish between clot and blood to reduce blood loss as well as the clot-dissolving drugs need.
It also allows physicians to focus on optimising thrombus removal procedures.
High power and low-profile catheter, CAT 7 includes laser-cut hypotube technology and circumferential sweep for maximal clot extraction and dependable delivery.
Penumbra stated that CAT 12 is a large-lumen aspiration catheter which features laser-cut hypotube-based catheter engineering that enables advanced deliverability and torque-ability in the body.
Penumbra Europe, Middle East and Africa region vice-president and head Joan Kristensen said: “Blood clots in the body can be difficult to access and are potentially life-threatening. Until now, treatment options have been limited.
“The introduction of our Lightning portfolio in Europe will expand access of our most advanced clot removal technology for the body, which combines intelligent aspiration and innovative catheter engineering to remove blood clots in a single session.
“With Lightning, mechanical aspiration technology continues to advance to meet the needs of patients, which is our core purpose at Penumbra.”



