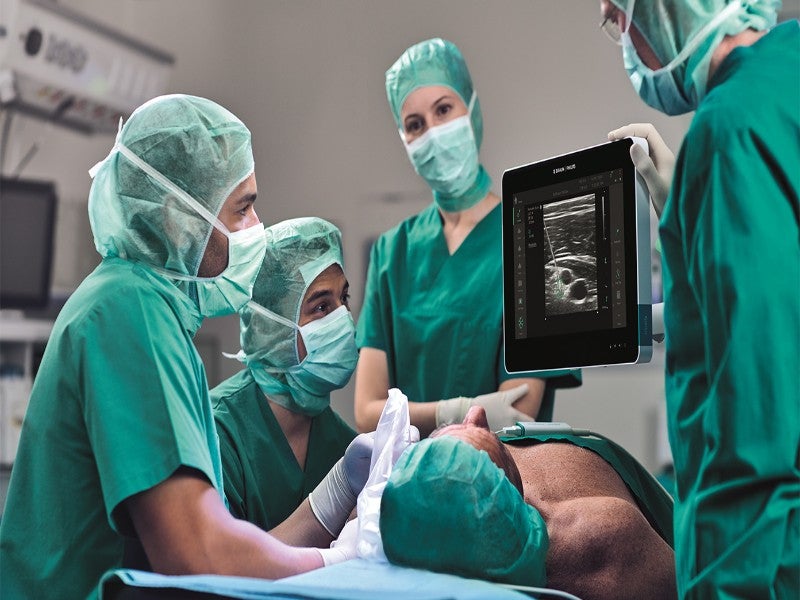
Royal Philips and B. Braun have received 510(k) clearance from the US Food and Drug Administration (FDA) for Onvision, an ultrasound guidance solution for real-time needle tip tracking during regional anaesthesia.
Onvision is designed to assist anesthesiologists in precisely placing the needle tip inside the body for peripheral nerve blocks (PNBs) by accurately indicating the position of the needle tip, both in and out of the ultrasound viewing plane.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
Additionally, it is said to reduce procedural time by enabling the user to align the needle with the probe in a user-friendly interface.
Accurate needle placement is required for the success of regional anaesthesia procedures, including PNBs.
Onvision solution, along with a dedicated Stimuplex Onvision needle, is available exclusively on the latest version of the B. Braun and Philips Xperius ultrasound system.
Philips Innovation for Image Guided Therapy head Tobin Taylor-Bhatia said: “The increased confidence and predictability offered by the Xperius Ultrasound System and Onvision needle tip tracking is empowering more anesthesiologists to embrace regional anaesthesia as a viable and effective alternative to general anaesthesia.

US Tariffs are shifting - will you react or anticipate?
Don’t let policy changes catch you off guard. Stay proactive with real-time data and expert analysis.
By GlobalData“By innovating together with B. Braun, we have created a solution to one of the biggest challenges in regional anaesthesia – accurate positioning of the needle tip in the body.”
FDA clearance for Onvision follows a multi-year strategic partnership between Philips and B. Braun, focusing on innovation in ultrasound-guided regional anaesthesia.
Braun Medical Affairs corporate vice-president Dr Angela Karpf said: “Onvision is the groundbreaking technology that allows anesthesiologists the predictability and confidence they need while positioning the needle during a procedure.
“B. Braun continues to add products to our regional anaesthesia therapy portfolio that help achieve faster post-surgical recoveries, optimise procedural workflow and enhance user experience.”
Onvision solution, which is also CE-marked, is currently available in the EU and in Chile. It is expected to be available in the US by the end of this year.





