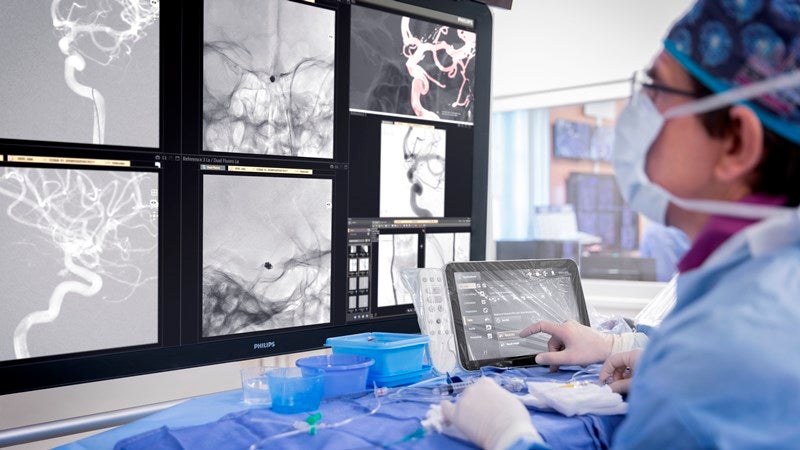
Royal Philips (Philips) has announced a clinical study to evaluate the effect of a ‘Direct to Angio Suite’ workflow on outcomes of stroke patients.
WE-TRUST (Workflow optimisation to rEduce Time to endovascular ReperfUsion in Stroke Treatment) is a multicentre, prospective, randomised, controlled, open-label, blinded-endpoint study, which will evaluate if Philips’ advanced image-guided therapy platform can diagnose, plan and treat stroke patients in the interventional suite without needing an initial CT or MRI exam.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
The trial will begin in the first half of 2020, with expected completion in 2022. It will be carried out across eight locations and will involve more than 460 patients across the world.
For stroke patients, outcomes are linked to how quickly treatment is carried out. A delay of every 30 minutes reduces the possibility of a positive outcome by 14%, while every hour ages the brain by 3.6 years against a healthy ageing brain.
Several studies have shown that a Direct to Angio Suite workflow can not only cut down the time to treatment but also boost patient outcomes.
Royal Philips is developing technology that can boost the CT-like images of the brain created with the X-ray system in the suite.
The trial is expected to offer a thorough assessment of the impact of this technology and workflow innovation on time-to-treatment and neurological outcomes in patients.
Philips image-guided therapy systems general manager Ronald Tabaksblat said: “With extensive clinical research demonstrating the benefit of a treatment approach that combines thrombectomy and clot-busting drugs, stroke patient triage and treatment has changed dramatically in recent years.
“For stroke patients, ‘time is brain’. The WE-TRUST trial will assess the impact of a streamlined Direct to Angio Suite workflow on patient outcomes and has the potential to make a significant impact in this rapidly advancing field.”
The trial’s primary endpoint is the cognitive function in patients at three months following the procedure.





