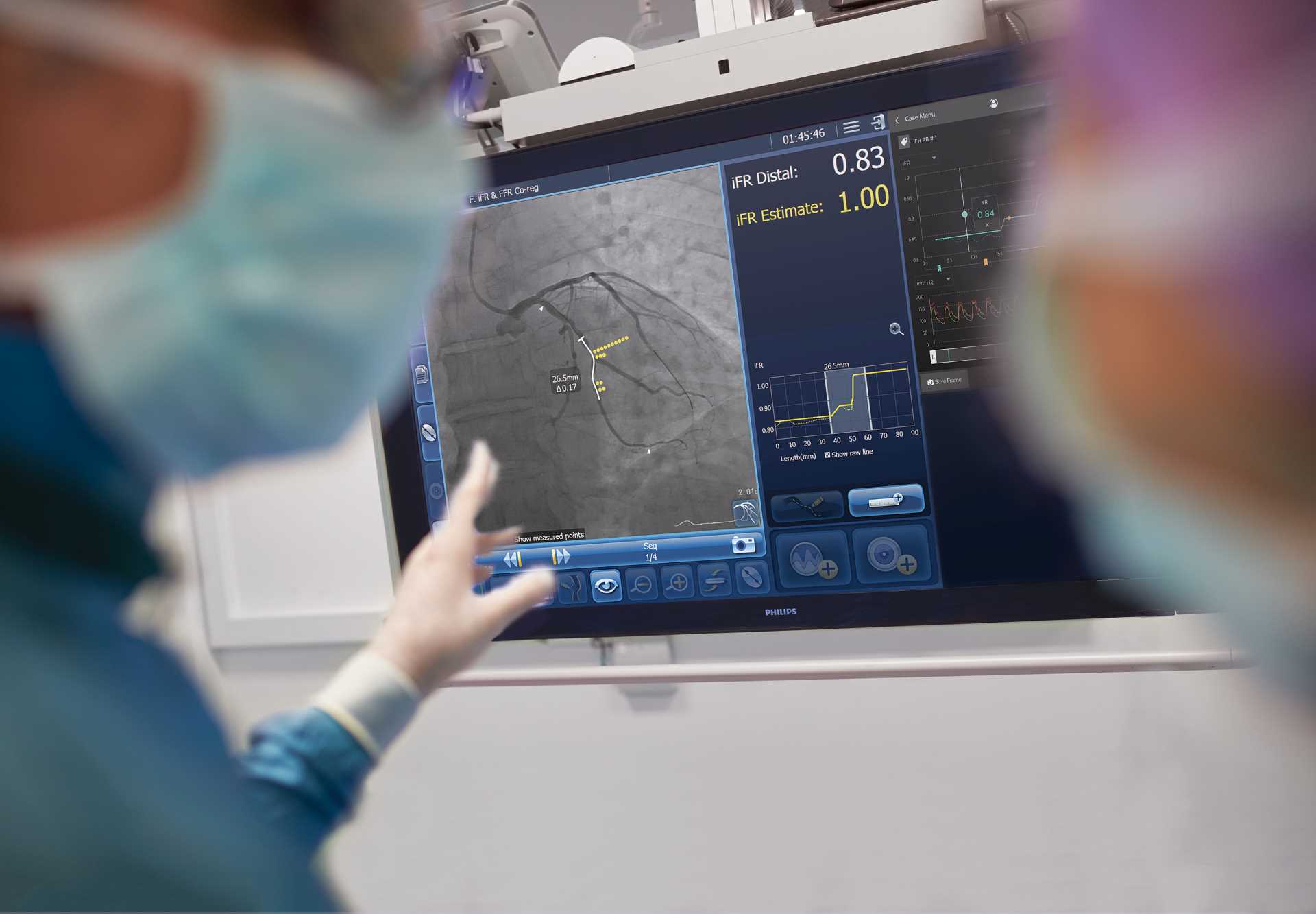
Royal Philips has commenced a study to analyse if the instantaneous wave-free ratio (iFR)-guided data co-registered on the angiogram could improve percutaneous coronary intervention (PCI) procedures to open blocked coronary arteries.
The Distal Evaluation of Functional performance with Intravascular sensors to assess the Narrowing Effect: Guided Physiologic Stenting (DEFINE GPS) study has enrolled the first subject at St Francis Hospital, New York, US.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
A global, multi-centre, prospective, randomised, controlled and adaptive trial, DEFINE GPS will enrol a total of 3,200 subjects at 100 sites globally.
The goal of the trial is to show enhanced outcomes and increased cost-effectiveness with PCI procedures guided by iFR measurements along with Philips image-guided co-registration system, SyncVision, compared to the existing standard of care of angiogram alone.
Royal Philips image guided therapy devices senior vice-president and general manager Chris Landon said: “iFR continues to be adopted in clinical practice, with mounting evidence that this innovative technology contributes to improved outcomes, reduced costs and enhanced patient experiences.
“This major study will provide a definitive answer to the question of whether a functional guidance strategy throughout the procedure demonstrates superior outcomes and reduces costs.”
In the study, an iFR pullback measurement will be placed on the angiogram to offer more accurate data on the region which needs treatment within the vessel.
iFR pullback measurement utilises pressure wires to locate the physiological profile of disease distribution along the affected vessel’s length.
Furthermore, the study will leverage iFR data to analyse if the treatment can effectively restore adequate blood flow to avert ischemia or if additional therapy is required.
Philips noted that the latest trial is a follow-up to a one-year DEFINE PCI study, which analysed a similar method to potentially treat residual ischemia to enhance clinical outcomes in coronary stent patients.
An image-guided, relatively less invasive treatment, PCI is performed to open a coronary artery blockage that leads to decreased blood flow to heart tissue.
Last week, Philips voluntarily recalled breathing devices and ventilators after it found possible health risks linked to the polyester-based polyurethane sound abatement foam used in these products.





