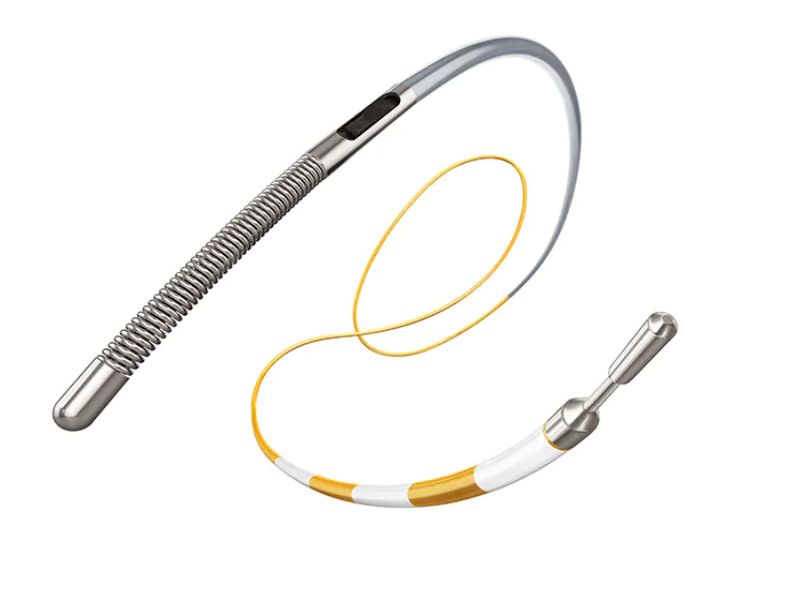
Royal Philips has launched a solid core pressure wire, called OmniWire, for coronary artery interventional procedures.
The solid core construction of the wire enables physicians to operate it in the patient’s circulatory system to measure blood pressure down the vessel and guide the delivery of catheters and stents.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
Furthermore, OmniWire is designed to assist the instant wave-free ratio (iFR) and fractional flow reserve (FFR) measurements.
It also integrates with the company’s IntraSight interventional applications platform, which is developed to co-register iFR data onto the angiogram to precisely detect the parts of vessels that need treatment.
Philips Image Guided Therapy Devices senior vice-president and general manager Chris Landon said: “With integration and co-registration on our IntraSight platform, measurement with iFR, and now enhanced wire performance, thanks to OmniWire, we are providing clinicians with an advanced solution at every step of the procedure.
“Physicians can confidently use a functional guidance strategy across all their patients, including in complex and multi-vessel cases. Today’s announcement demonstrates how our unique portfolio of systems, smart devices, software and services combines to deliver advanced procedure-oriented solutions.”
Typically, pressure wires use a hollow metal tube to cover the wiring that transmits the pressure information. Because of their thin walls, these wires are prone to damages during the procedures.
Philips’ OmniWire uses enhanced conductive ribbons embedded in its outer polymer layer to communicate pressure information.
The front part of the wire is made from a super-elastic, durable material called Nitinol, while a high-strength cobalt alloy has been used for the construction of the proximal end.
OmniWire, which has received clearance from the US Food and Drug Administration (FDA) and approval from the Japan Pharmaceuticals and Medical Devices Agency, is currently available in the US and Japan.
Last month, Philip’s entered a strategic collaboration with BioIntelliSense to enhance remote monitoring for at-risk patients at home.





