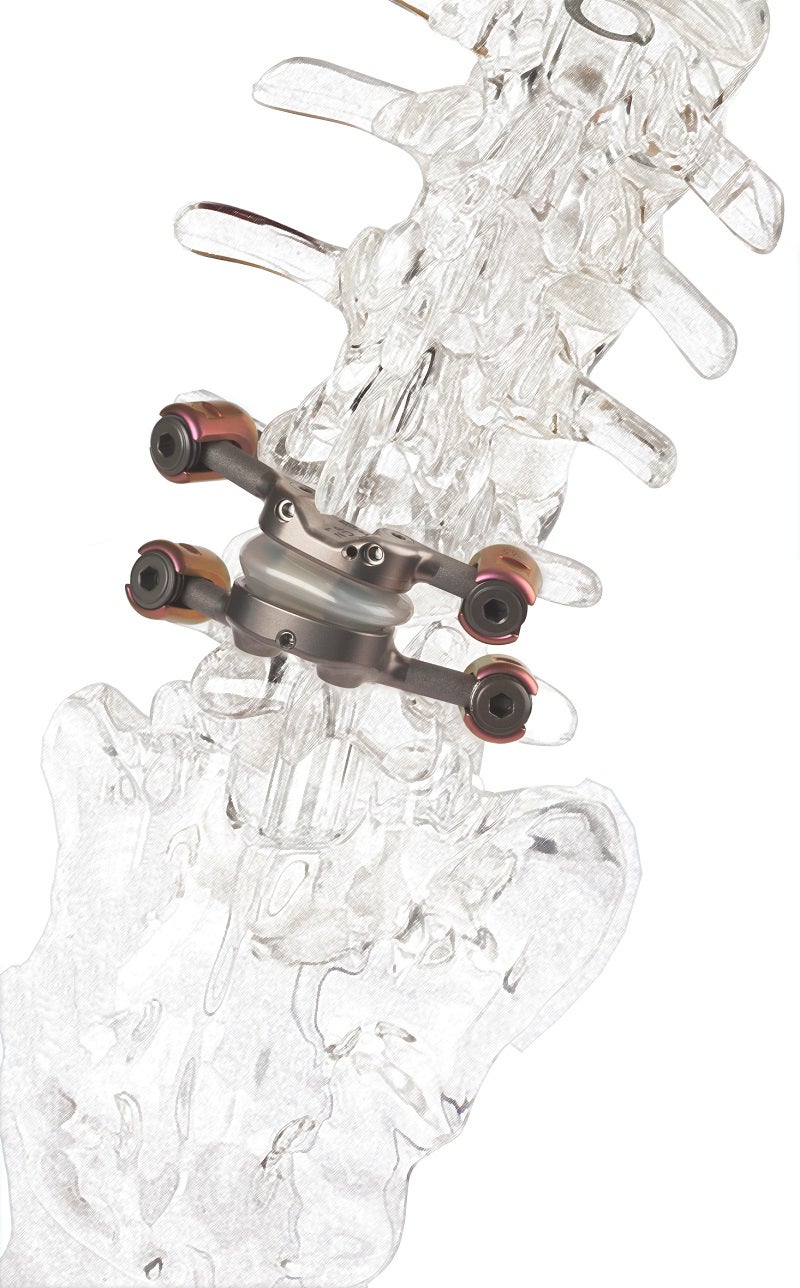
Medical technology firm Premia Spine has reported positive data from a single-centre study of its TOPS System for the treatment of lumbar spinal stenosis and degenerative spondylolisthesis.
The study was published in the Operative Neurosurgery journal by authors Werner Lack, Hans Paul Kutschera and Josef Krugluger under the title ‘Mobility-Maintaining Arthroplasty of the Lumbar Spine with the Second-Generation TOPS System: A Case Series’.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
The authors concluded that the TOPS facet system has the capability to relieve pain in the legs and back while maintaining close-to-normal range-of-motion (ROM) without inducing adjacent segment disease (ASD) over a time period of four years.
According to the authors, the system is the first that enables patients to settle into their natural sagittal balance.
This offers new opportunities for non-fusion treatment of degenerative spondylolisthesis and spinal stenosis with mobility-maintaining dorsal instruments.
Premia Spine stated that the study described a single-centre experience using a facet replacement implant that stabilises the spine, prevents further degeneration and preserves ROM.
In the study, 17 patients who received the implant after a laminotomy were assessed for improvements in ROM and visual analogue scale (VAS) over an average time period of 51 months.
With an average improvement of 81%, the average decline in VAS score was 7.5 reported at three months, 6.8 at 12 months and 6.7 at the longest follow-up.
The average ROMs at the index level were 8.2 degrees preoperatively and 7.4 degrees postoperatively.
Premia Spine CEO Ron Sacher said: “We are encouraged by these long-term results, which support other studies that demonstrate the potential of the TOPS System as a viable alternative to fusion for the treatment of spinal stenosis and degenerative spondylolisthesis.
“Our goal remains to deliver an implant that allows patients to retain the natural motion of their spine with less pain.
“These data bolster our case for TOPS as a new treatment paradigm for patients suffering from these diseases.”
The TOPS System has been designed to provide stability, durability and mobility following decompression for lumbar spinal stenosis and degenerative spondylolisthesispatients.
Last year, the US Food and Drug Administration (FDA) granted Breakthrough Device Designation to the TOPS System.





