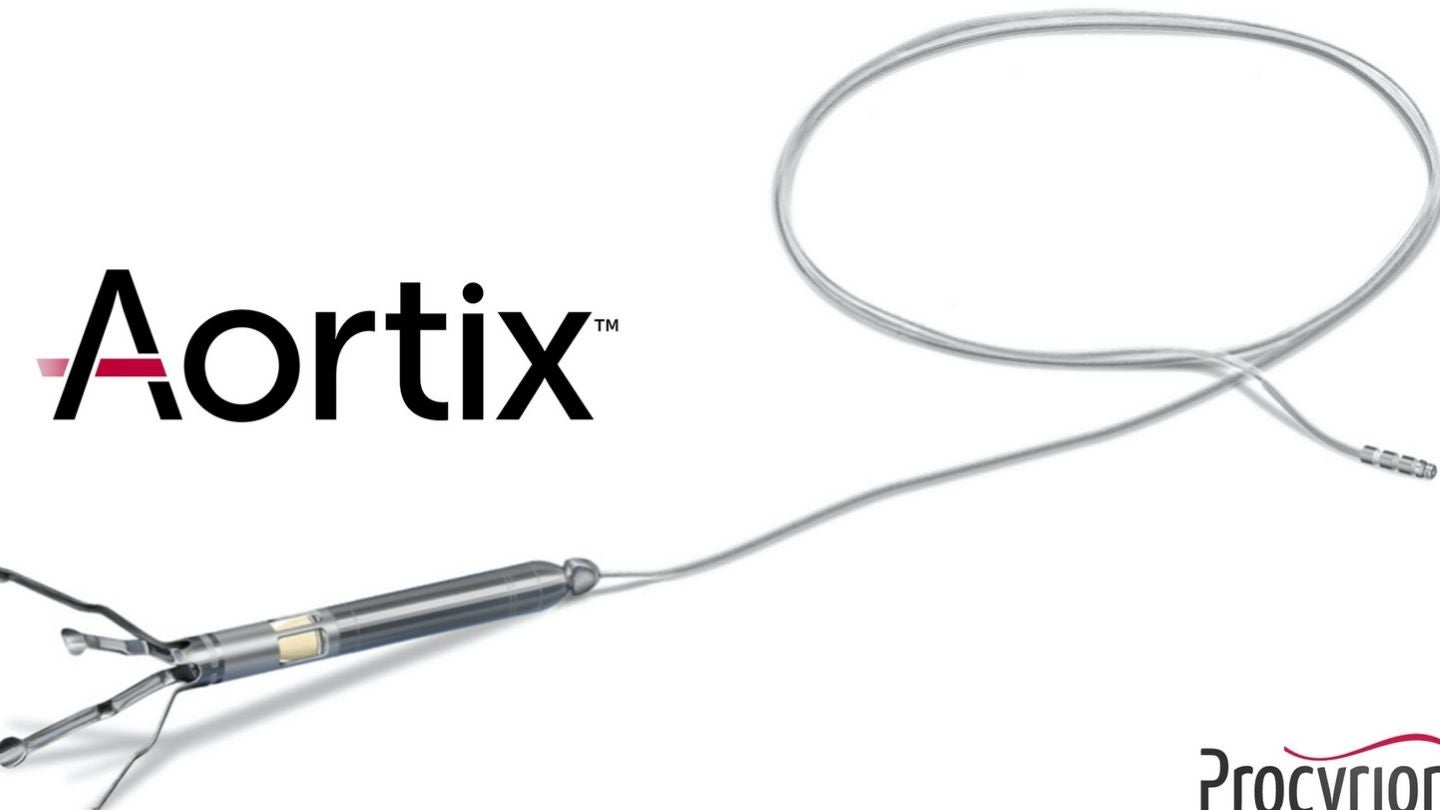
Procyrion has treated the first patients with its Aortix percutaneous mechanical circulatory support (pMCS) device in the A PRIORI (aortix therapy for perioperative reductIon of renal injury) pilot study.
The trial has been designed to assess the Aortix device’s ability to prevent acute kidney injury (AKI) during cardiac surgery.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
Procyrion will recruit up to 50 patients for the study across four clinical sites in Australia, namely Royal Adelaide Hospital, Monash Health, Princess Alexandra Hospital and Macquarie University.
At the start of the scheduled surgical procedure, the Aortix pump is placed in the descending thoracic aorta through a percutaneous catheter procedure.
It helps to directly enhance perfusion to the kidneys while lowering cardiac energy requirements, stated the company.
The set-up of the pump can be completed within 15 minutes, and the intra-aortic placement helps avoid the risk of device-related thrombotic stroke or damage to the heart or valves.

US Tariffs are shifting - will you react or anticipate?
Don’t let policy changes catch you off guard. Stay proactive with real-time data and expert analysis.
By GlobalDataThe device’s design is said to leverage fluid entrainment to directly enhance renal artery flow and pressure, as well as enable physiologically natural therapy delivery.
According to the company, the Aortix pump should remain in place for up to 48 hours post-surgery to maintain increased renal perfusion.
This provides additional protection against postoperative risk factors for AKI, in addition to decreasing the workload for the recovering heart.
Procyrion president and CEO Eric Fain said: “The initiation of the A PRIORI study, after recently completing our pilot study in treating cardiorenal syndrome patients, is an exciting step that contributes to the growing body of evidence demonstrating the potential of the Aortix pump technology to improve outcomes for patients with cardiac and renal impairment.”





