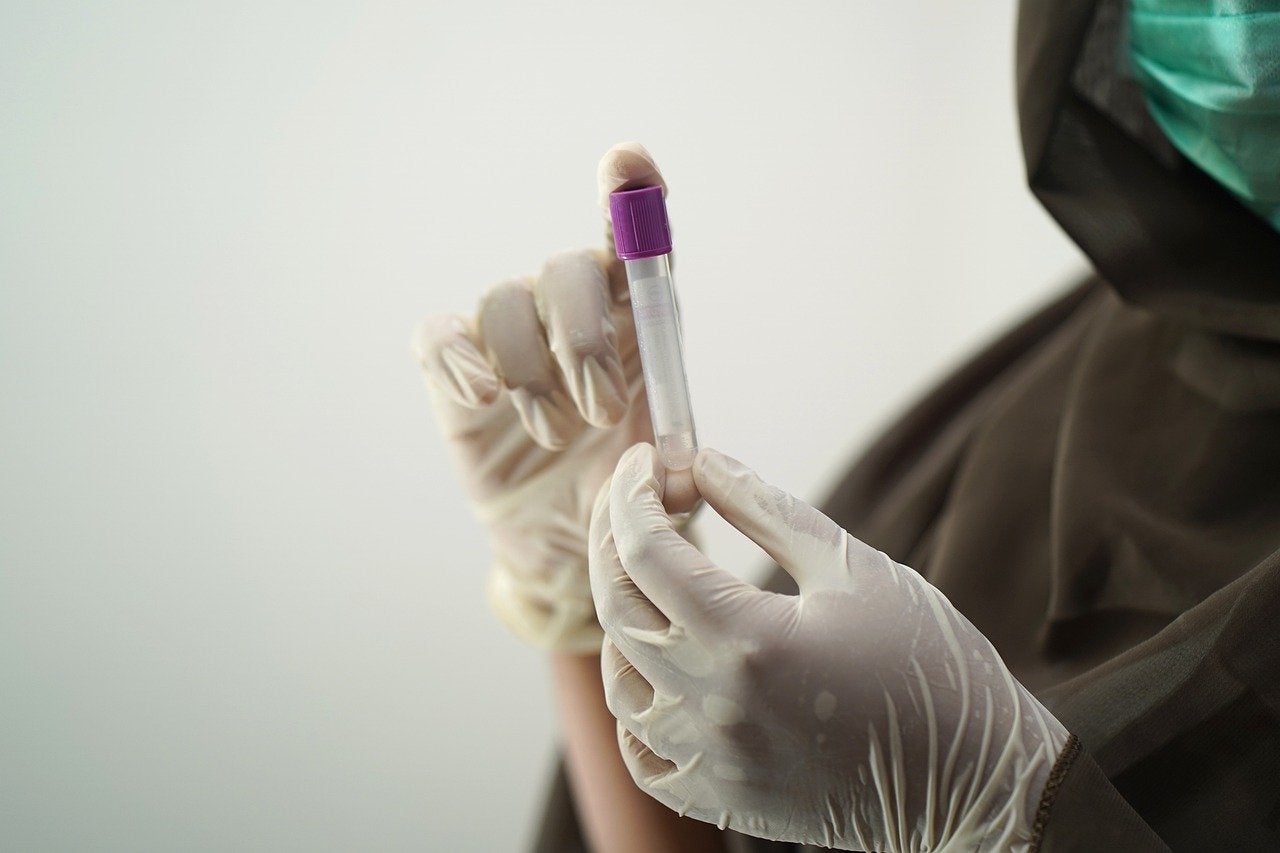
Purlab has launched an at-home saliva-based Covid-19 testing solution for individuals, companies and schools to make testing easy and accessible.
The technology can potentially solve the concerns of test result turnaround time, efficiency and mass testing to provide a user-friendly process.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
The test kit costs $145, which is inclusive of a collection device, doctor fees, lab processing fees, technology and shipping.
On receipt of the test kit to their home, users can register it through the Purlab app or on Purlab.com and scan the barcode on the tube.
They can take the saliva test by spitting into the tube provided with the kit, pack it in a pre-labelled box and then drop it in the mail in minutes. Users receive test results from the lab in 24 hours through email and text.
Purlab president Michael Cohen said: “Purlab is making it easier than ever to get tested for Covid-19 and receive accurate results in a timely manner.

US Tariffs are shifting - will you react or anticipate?
Don’t let policy changes catch you off guard. Stay proactive with real-time data and expert analysis.
By GlobalData“We have the most user-friendly Covid-19 test on the market today and the only one with a mobile app, allowing for a seamless process for people in the comfort of their own homes.”
The test is developed and run in partnership with Rutgers University RUCDR Infinite Biologics Lab and is the first SARS-COV-2 Saliva test to receive Emergency Use Authorisation (EUA) from the US Food and Drug Administration (FDA).
In a separate development, molecular diagnostics firm Co-Diagnostics has completed principal design work for a PCR test using which researchers can use to detect certain mutations in a newly detected variant of SARS-CoV-2 known as VUI 202012/01.
Co-Diagnostics CEO Dwight Egan said: “One of the most important advantages of our CoPrimer platform is its ability to reliably and accurately differentiate between similar genetic sequences, in order to reduce the likelihood of a false-positive diagnosis.
“The multiplexing capabilities of our CoPrimer platform go hand-in-glove with the technology’s applications in detecting small genetic variations in a given gene.”





