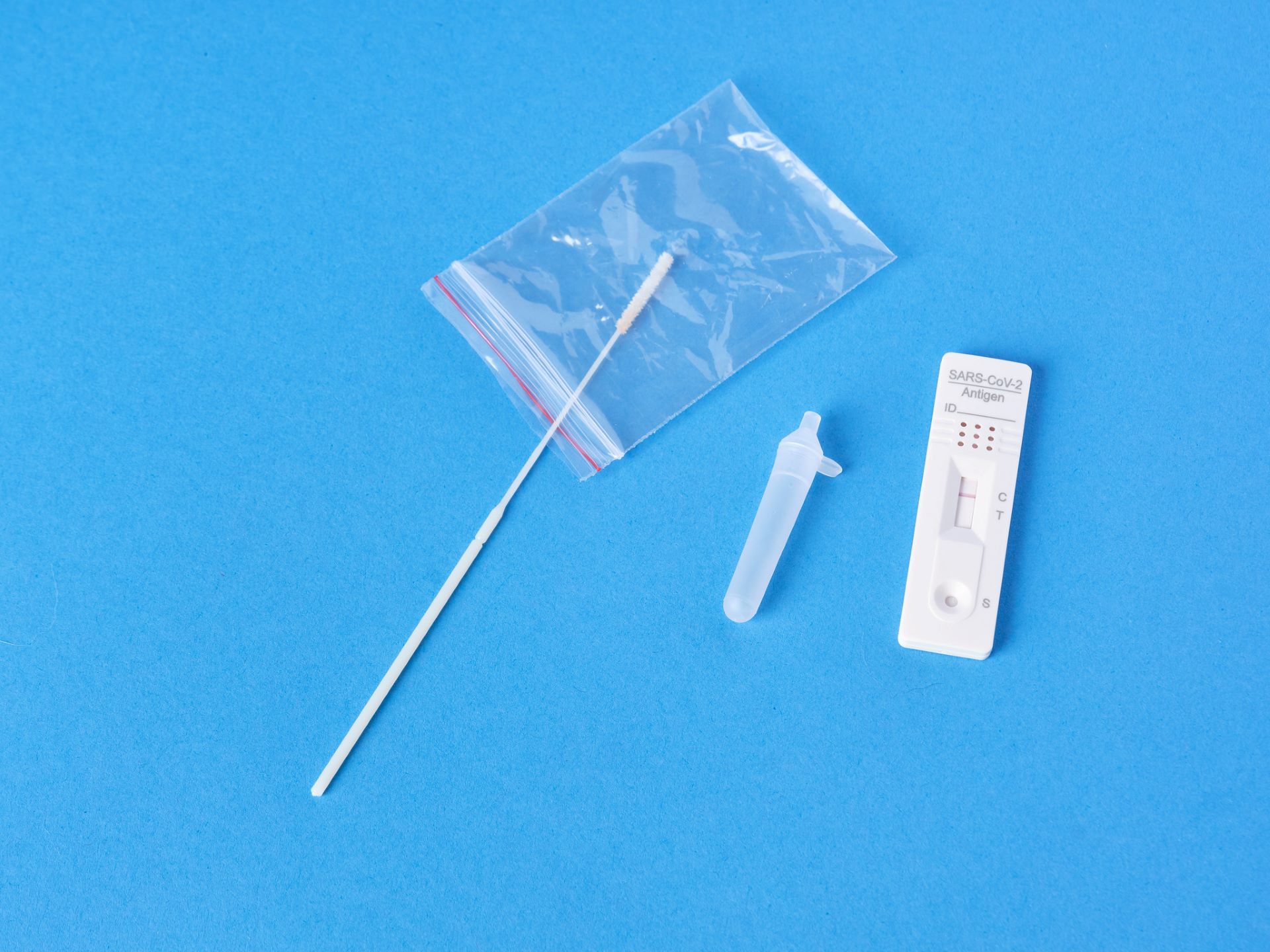
The US Food and Drug Administration (FDA) has granted an emergency use authorization (EUA) for Quidel’s new QuickVue At-Home OTC COVID-19 Test to be marketed in the country.
The test will be used for the qualitative detection of the nucleocapsid protein antigen from SARS-CoV-2 from people with or without symptoms.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
It can also be used in cases where Covid-19 is suspected for other epidemiological reasons. Individuals should be tested twice across two or three days with a gap of at least 24 hours between tests.
The QuickVue test is authorised for non-prescription home use with self-collected direct anterior nares samples from people aged 14 years and above or with adult-collected specimens from children aged two years and above.
Quidel president and CEO Douglas Bryant said: “Studies are confirming that serial testing with rapid antigen tests is a crucial resource for people to know their current health status and make prudent decisions to protect themselves, their loved ones and their communities.”
The test provides results in agreement with PCR 83.5% of the time when positive and 99.2% when negative.
In a separate development, the US FDA has granted EUA to Becton, Dickinson and Company’s (BD) rapid antigen test which can be used for SARS-CoV-2 screening through serial testing of asymptomatic people.
The BD Veritor Plus System for Rapid Detection of SARS-CoV-2 can qualitatively identify SARS-CoV-2 nucleocapsid antigens from direct anterior nasal swabs in Covid-19 suspected people within five days of symptom onset.
The EUA also supports SARS-CoV-2 screening through serial testing of asymptomatic people when tested twice across two or three days with a gap of at least 24 hours between tests.
Serial testing on the BD Veritor Plus System can be performed in any setting with a CLIA certificate of waiver.
BD Life Sciences president Dave Hickey said: “Frequent testing of individuals without symptoms will enable those with negative results to resume their normal school or work routines and will help to identify and isolate positive cases of COVID-19 as early as possible to prevent further spread.”
Last month, the US FDA granted EUA to BD’s new rapid antigen test that can identify SARS-CoV-2, influenza A and influenza B in a single test.





