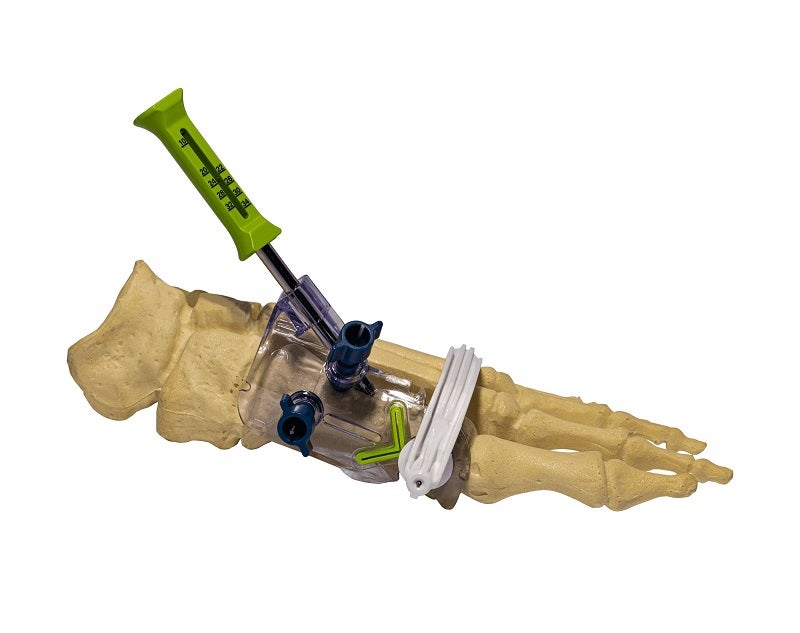
RELJA Innovations has introduced the new minimally invasive surgery (MIS) Precision Chevron Bunion System for osteotomy.
The new system allows surgeons to perform a mild to moderate chevron bunion osteotomy, as well as subsequent osteotomy fixation, using a minimally invasive surgical technique.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
It comprises a single pre-sterilised part number, which includes the implants and instruments required for the procedure.
The company stated that the same kit can be used for the left or right foot, as well as on feet of any size.
Fixation is achieved by a 3.5mm diameter cannulated screw, and the two screw lengths included were shown to provide adequate fixation for all foot sizes.
RELJA founder and CEO, board-certified foot and ankle surgeon Dr Robby Amiot said: “The MIS Precision Chevron Bunion System allows me to complete a chevron osteotomy with minimally invasive technique in the same or less time than an open procedure.
“In addition, my patients have requested less opioids than a traditional open chevron procedure and have expressed great satisfaction.”
With the new MIS Precision Chevron Bunion System, surgeons will be able to perform a traditional and biomechanically stable chevron osteotomy using a surgical incision as small as 1cm.
This minimises the disruption of soft tissue and is expected to produce beneficial results similar to other MIS systems, RELJA said.
Created by the company in partnership with consulting surgeons, the new system is now available for commercial use in the US.





