Biomedical engineers from Emory University and Georgia Institute of Technology have developed a miniature self-sealing model system for studying the bleeding and clotting of wounds. The device could be used as a drug discovery platform and potential diagnostics tool.
Blood clotting involves damaged blood vessels, platelets, blood clotting proteins which form a net-like mesh and the flow of the blood itself. It normally takes around eight minutes for blood flow into a wound to stop. Endothelial cells play a crucial part in this; without them clotting would not occur.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
“Current methods to study blood clotting require isolation of each of these components, which prevents us from seeing the big picture of what’s going with the patient’s blood clotting system” said William Lam, lead author of the study and assistant professor in the Department of Pediatrics at Emory University School of Medicine and in the Wallace H. Coulter Department of Biomedical Engineering at Georgia Tech.
This new blood clot model system is an advancement on current methods because it is the first to include all the aspects of blood vessel injury seen in the microvasculature. These aspects include blood loss due to trauma, clot formation by whole blood and repair of the blood vessel lining. However, the model does not include smooth muscle and does not reproduce aspects of larger blood vessels.
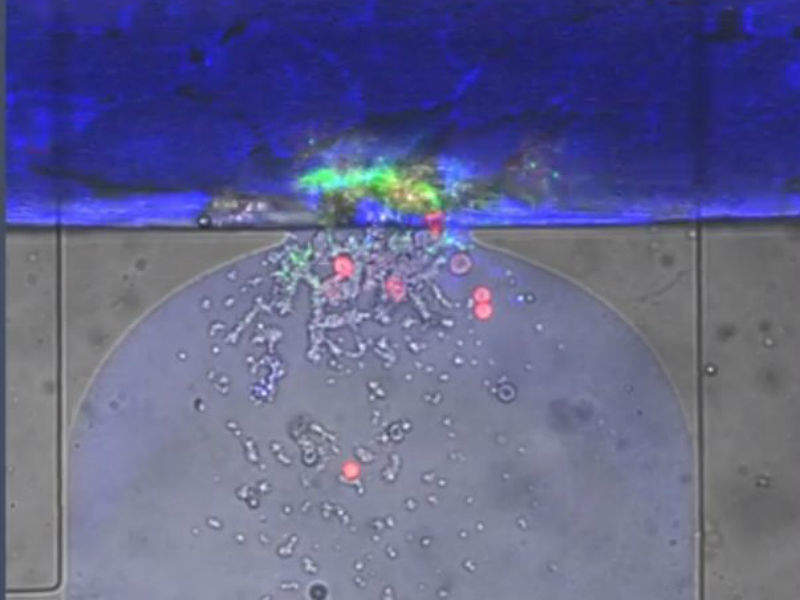
The system consists of a layer of human endothelial cells, which line blood vessels, cultured on top of a pneumatic valve. The ‘wound’ is created by activating the pneumatic valve, opening what the researchers have described as a ‘trap door’. Donated human blood flows through the wound, which is about 130 micrometres across.
Drugs and other factors were used to manipulate the system so that it replicated blood clotting disorders. For example, blood from haemophilia A patients was used to form abnormal clots and showed extended bleeding times in the model.
The study has been published in a Nature Communications paper in which the authors also describe how the drug eptifibatide affects the interactions of platelets and other cells in the 3D space of a wound.
The full video of the bio-mechanical wound clotting process is available at: https://youtu.be/l7k1dGfKG0g





