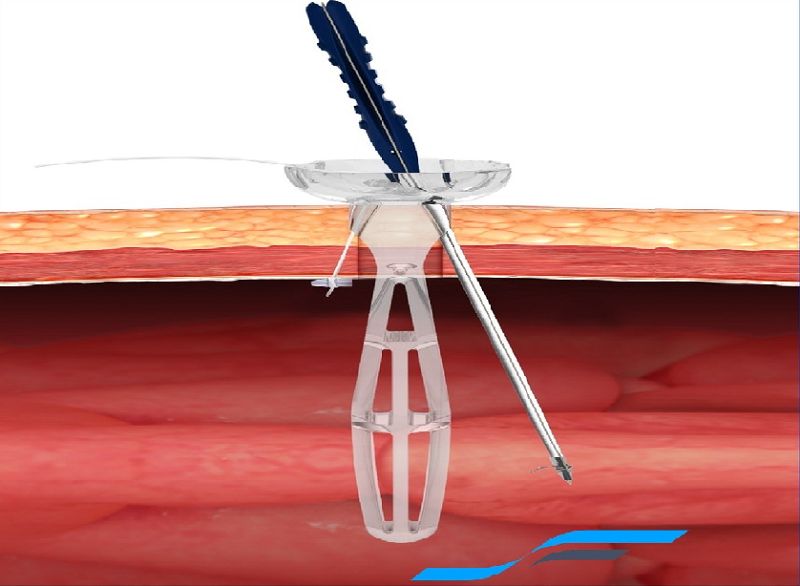
Sharp Fluidics has announced the acquisition of laparoscopic and robotic port site closure technology neoClose from neoSurgical for an undisclosed sum.
neoClose enables surgeons to close port site defects, up to 3cm, following laparoscopic and robotic abdominal surgery.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
The technology uses bio-absorbable anchors with a pre-attached suture and is changing the standard of care in port site closure. It is considered to be quicker and safer and gives less port site pain compared to standard practices of closing port sites.
Sharp Fluidics president Doug Rimer said: “We strive to introduce innovative thinking to surgical closure, and neoClose is highly complementary to our Operative Armour System.
“With Operative Armour, clinicians can self-dispense and self-secure their own suture needles in open surgery closures without scrub tech assistance, dramatically improving workflow efficiency while minimising exposure to contaminated suture needles. neoClose enables us to now offer an extremely elegant and user-friendly, needleless approach to perform safer, more efficient closure of minimally invasive surgeries.”
The acquisition of neoClose is expected to expand Sharp Fluidics’ product portfolio. The company’s Operative Armour System leads the way in open surgery closure efficiency and safety.
American Association of Hip and Knee Surgeons orthopaedic surgeon and former president Mark Froimson said: “neoClose and Operative Armour reduce OR surgical times and improve OR staff safety, two of the most important initiatives that many leading hospitals have prioritised.”
neoClose is neoSurgical’s initial product and is approved in the US and Europe for commercial sale.
Surgical device company Sharp Fluidics specialises in developing and marketing medical technologies and devices to enhance surgical closure efficiency and safety.





