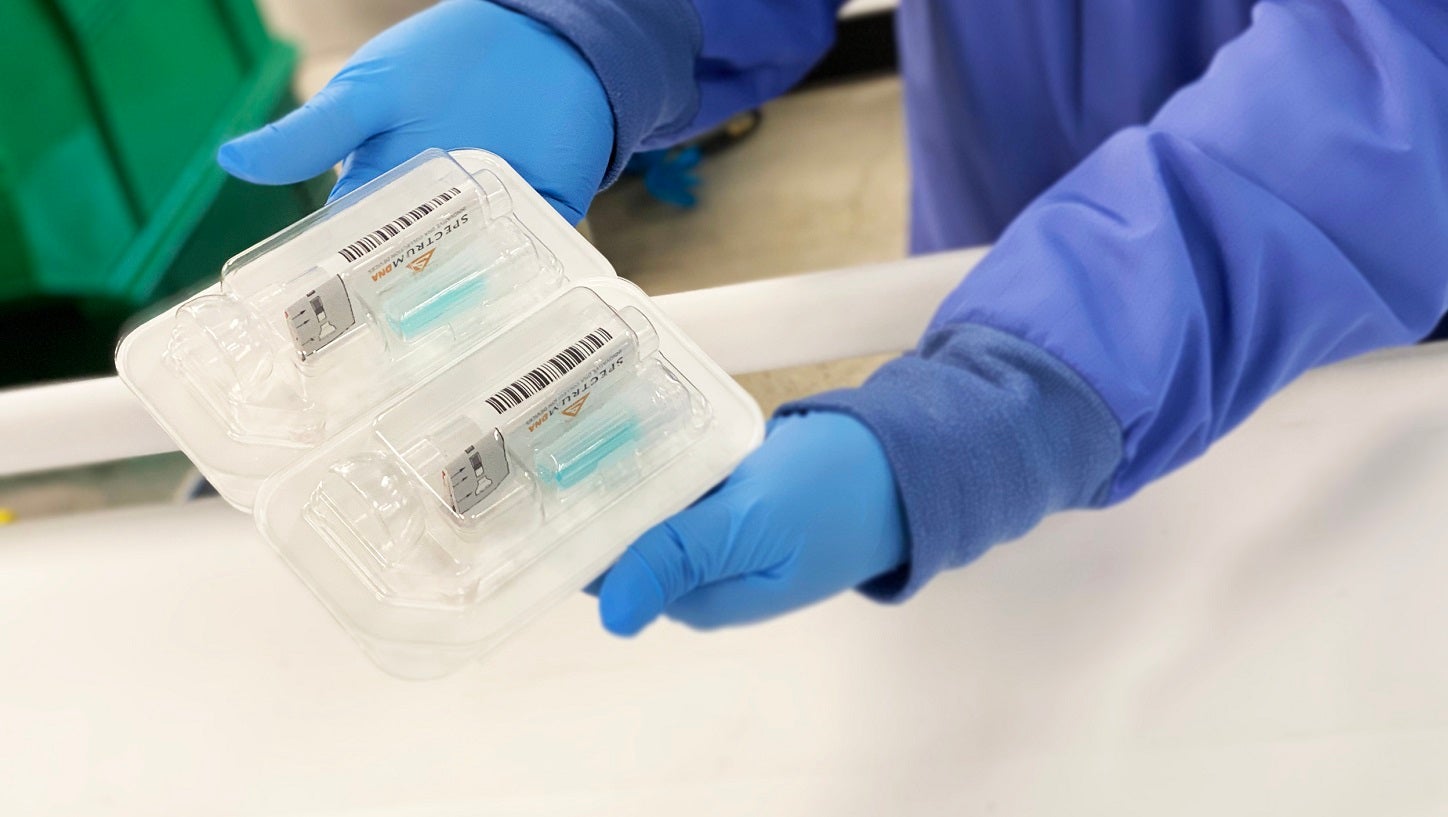
Spectrum Solutions has secured 510(k) Class II clearance from the US Food and Drug Administration (FDA) for its SDNA Saliva Collection Device.
Cleared as a microbial nucleic acid storage and stabilisation device, SDNA can maximise the detection of viruses at low levels. It also neutralises viruses within ten seconds of collection to alleviate unnecessary exposure.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
The device features a patented preservation media that keeps analytes stable at ambient temperatures for several weeks. This ensures safe, easy and secure specimen storage and transport.
Spectrum claims that its automated methods are carried out on industry-leading extraction and validation platforms, which ensures the highest degree of stringency and quality.
Spectrum Solutions CEO Stephen Fanning said: “This 510(k) clearance and certification as an IVD molecular diagnostic device enables physicians, hospitals, researchers and others to leverage our SDNA Saliva Collection Device in a broad array of FDA-approved and LDT diagnostic testing applications.
“Those conducting screening and diagnostic tests can confidently and safely expand access and opportunity incorporating saliva as a primary or alternative biomaterial into their existing testing protocols.”

US Tariffs are shifting - will you react or anticipate?
Don’t let policy changes catch you off guard. Stay proactive with real-time data and expert analysis.
By GlobalDataThe company received FDA Emergency Use Authorization (EUA) for its SDNA-1000 near the start of the pandemic.
After processing SARS-CoV-2 saliva tests for more than two years, the device and its patented nucleic acid preservation chemistry have been proven to have superior capabilities.
Spectrum Solutions chief medical officer Rohit Gupta said: “We couldn’t be more excited for the opportunity this new 510(k) device clearance delivers in supporting laboratory medicine and healthcare systems in safely and confidently decentralising specimen collections for precise and accurate testing.”





