Clinical stage bioelectric medtech firm SpineX has announced the enrolment of the first participant in a pivotal trial of the SCONE Device.
The SCONE trial has been designed to assess the effectiveness and safety of the device to treat neurogenic bladder.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
It is said to be the first large-scale pivotal trial to test a non-invasive spinal neuromodulation technology that has been developed to treat neurogenic bladder.
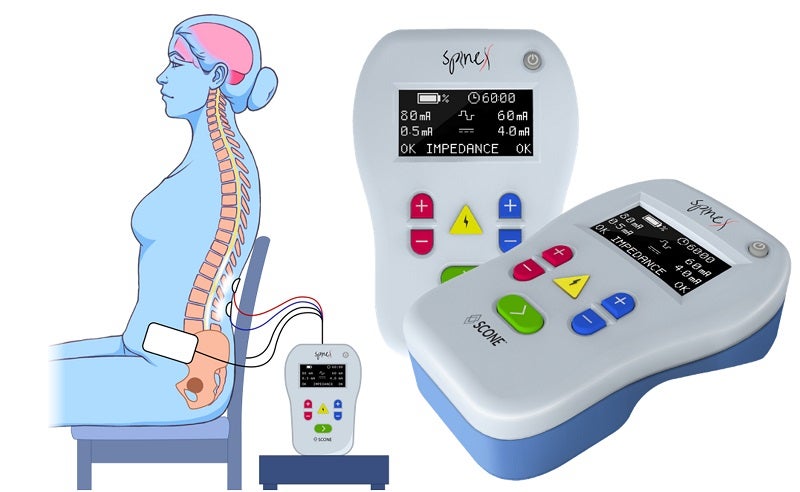
SpineX stated that the first participant was enrolled at Rancho Research Institute, Rancho Los Amigos National Neurorehabilitation Center’s research arm.
Neurogenic bladder is the most common comorbidity in people after paralysis.
Its effect goes beyond the inability to void when required or the necessity for daily repeating catheterisations to empty the bladder for the rest of one’s life.
People with neurogenic bladder will have symptoms like low capacity of bladder, lost sensation of bladder fullness, a constant fear of uncontrolled urine leakage, and frequent urination cycles during the day and night.
SpineX co-founder Dr Evgeniy Kreydin said: “For a person in a wheelchair, the inability to walk is the most obvious functional loss, but the impact on quality of life due to neurogenic bladder is unparalleled.”
The company said that the SCONE therapy has been designed to treat each of these symptoms.
The device provides proprietary modulated waveforms of changing currents to designated areas of the spine in a specific sequence through hydrogel electrodes directly over the skin.
Due to this, the user will not have any pain or discomfort.
SCONE is designed for the treatment of neurogenic bladder caused due to spinal cord injury, stroke, or multiple sclerosis.
SpineX CEO Dr Parag Gad said: “The initiation of the SCONE trial is an important milestone in bringing the world’s first non-invasive treatment modality for neurogenic bladder to market.
“We are committed to transforming bladder management into a catheter free and leak free world.”





