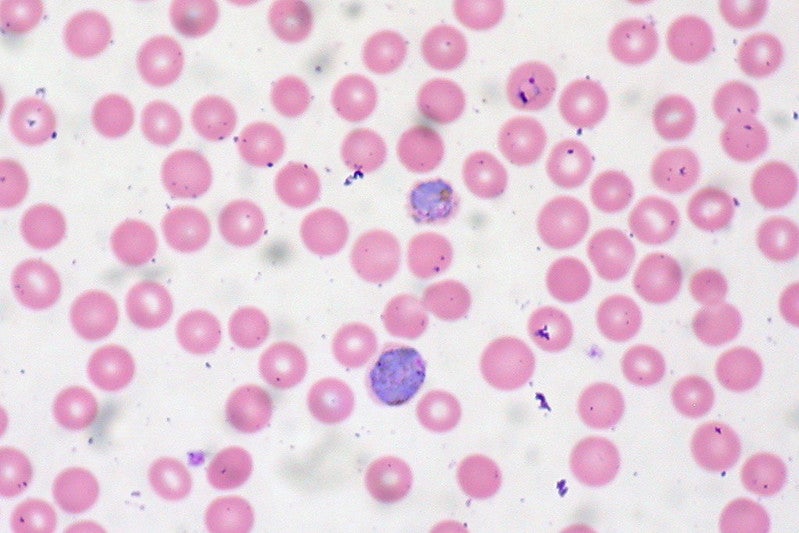
Sysmex has received marketing approval for its Automated Hematology Analyzer XN-31 for use in the diagnosis of malaria in Japan.
It is the first medical device for in vitro diagnosis to have been approved in Japan as a specially controlled medical device for use in the diagnosis of malaria (Class III).

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
The approval allows the device to be used for clinical applications as a measure against imported infectious diseases in Japan.
Automated Hematology Analyzer XN-31 is developed to help in the diagnosis of malaria by enabling qualitative analyses of formed elements in whole blood based on the count of DNA-containing malaria-infected RBCs (MI-RBCs).
The device uses electric impedance, light scattering by irradiating laser light onto cells that move inside a flow cell, or dye-binding for the analysis.
It counts, quantitated and identifies platelets, red blood cells and white blood cells. It also measures haemoglobin and determines hematocrit values, corpuscular constant, red blood cell distribution width and platelet distribution width.
Sysmex is planning to obtain marketing approval for the analyser in other countries in Asia and Africa.
Malaria is a protozoal disease, resulting from plasmodia transmitted by mosquitoes. It is considered to be one of the world’s three major infectious diseases.
Malarial parasites affect the circulating red blood cells (RBCs) in the body after a certain incubation period. High fever, headache, vomiting and anaemia are the symptoms associated with the diseases.
According to a report by the World Health Organization (WHO), at least 30,000 people get infected by malaria at their destinations every year and are diagnosed with the disease after returning home.





