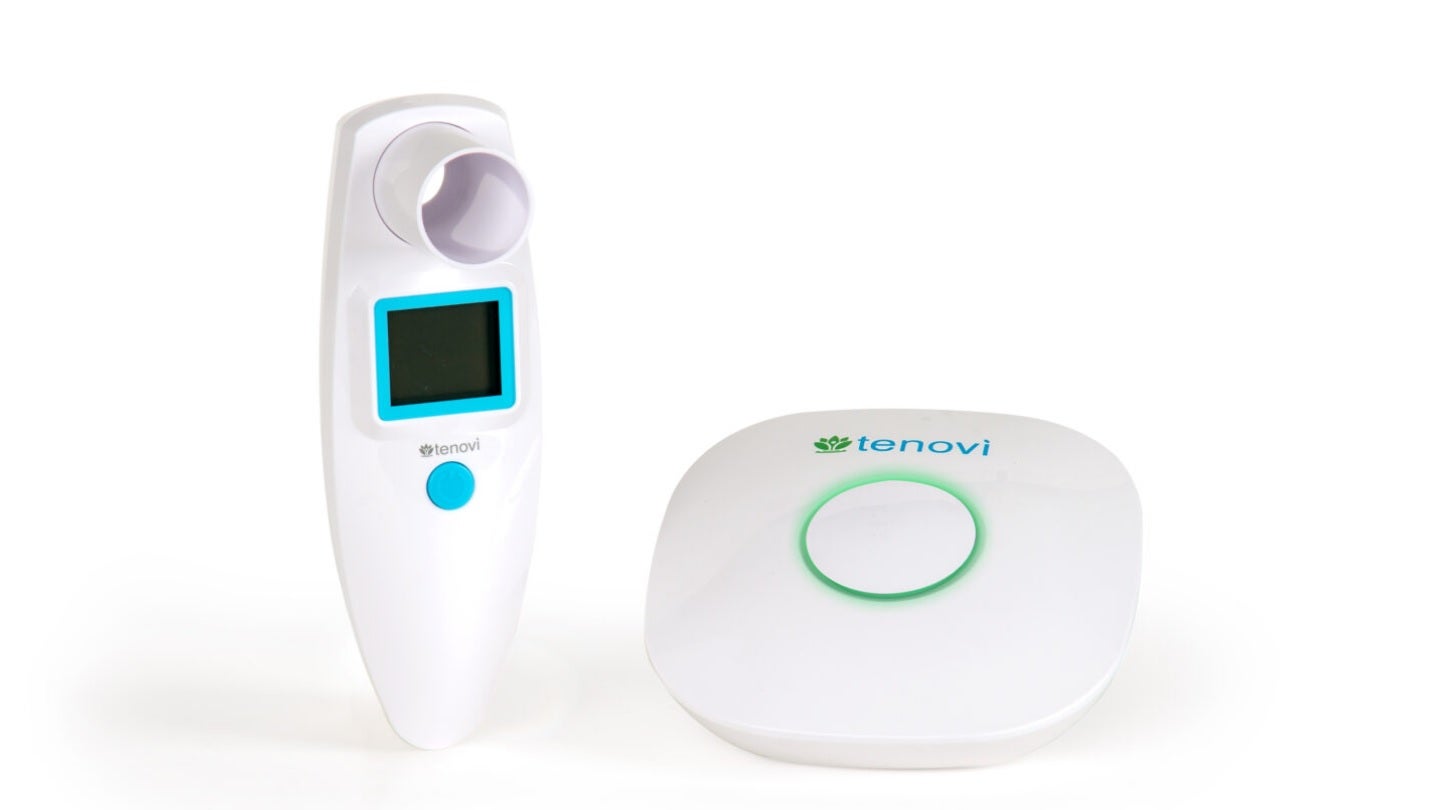
Remote patient monitoring (RPM) solutions provider Tenovi has introduced a new cellular-enabled remote peak expiratory flow meter (PFM) for asthmatic patients.
Traditional peak flow meters need manual recording and tracking of readings for the measurement of peak expiratory flow rate (PEF), an important indicator of lung function in asthma patients.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
The new device supports integration with the company’s Cellular Gateway to help asthma patients immediately transmit PEF and 1-second forced expiratory volume (FEV1) readings to clinicians.
The Bluetooth-enabled RPM device is designed to automatically send PEF and FEV1 to the Tenovi Cloud through the Tenovi Health Hub.
Designed to offer one-step, seamless recording, the device transmits data to a clinician portal to enable remote monitoring of patients.
According to the company, a recent study revealed that asthma patients with respiratory illness showed improved outcomes when FEV1 measurements were remotely monitored.
Tenovi founder and CEO Nizan Friedman said: “Today’s release of Tenovi’s cellular-based peak flow meter is a major milestone as it empowers physicians to provide better patient care and allows for significantly better patient adherence compared to the alternatives in the market.
“We are committed to continuing offering the most advanced RPM solutions to our telehealth, chronic care, software and RPM service company partners.”
Tenovi is a data aggregation and automation platform, which acts as a conduit between medical device manufacturers and RPM companies.



