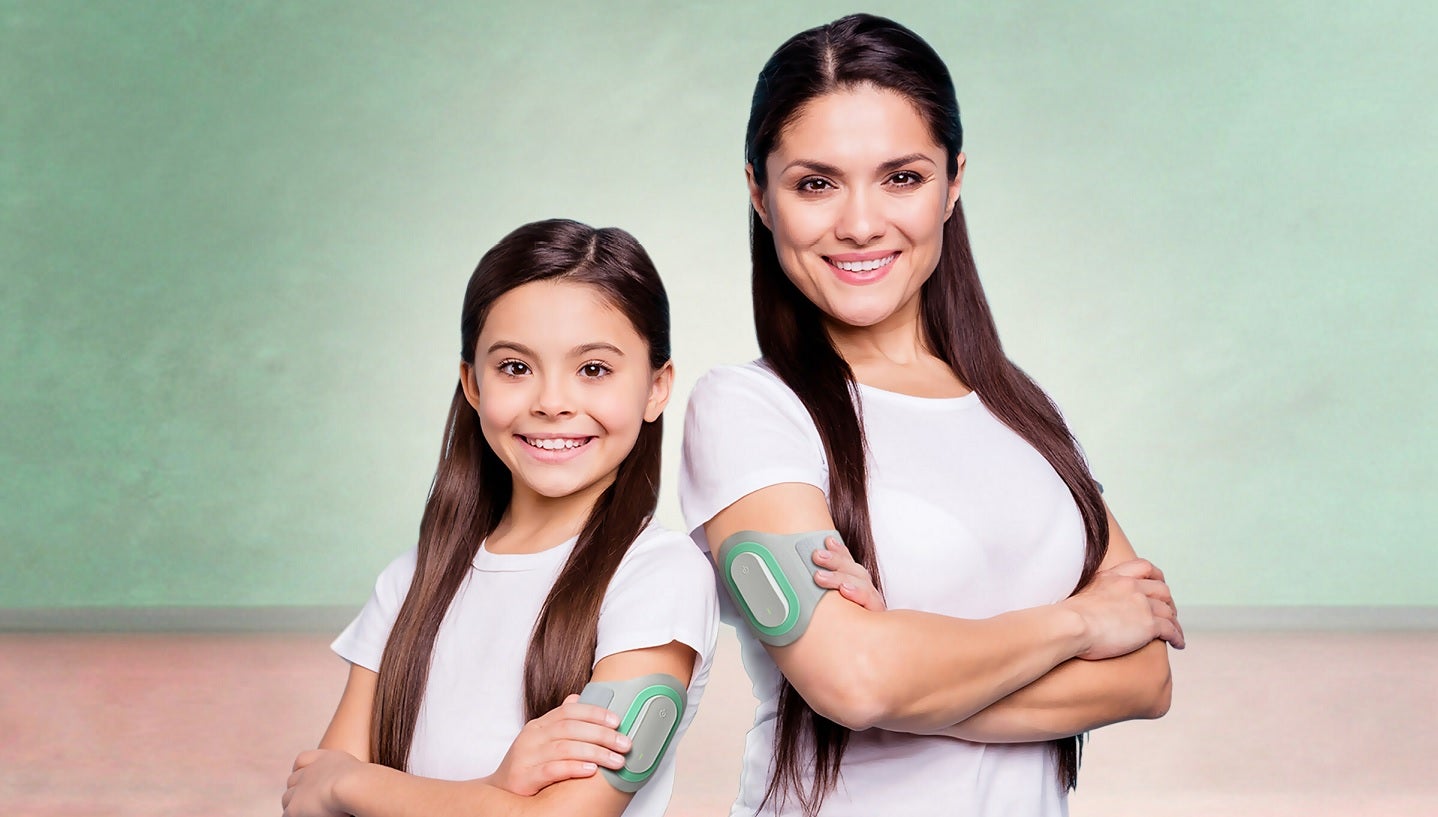
Theranica has secured clearance from the US Food and Drug Administration (FDA) for its Nerivio prescription wearable as a dual-use acute and preventive treatment for migraine.
Nerivio is a wearable migraine treatment that stimulates the pain receptors of the body to easily relieve both acute and chronic migraine pain.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
It is now indicated for migraine patients aged 12 years or above with or without aura.
The device had previously received approval as an acute treatment for migraine in the same patient group.
The approval was based on previous clinical trials, where Nerivio showed consistent relief of pain as well as associated symptoms of migraine.
Worn on the upper arm, the Nerivio device uses non-painful remote electrical modulation (REN) to activate the peripheral nerves.
This induces the internal pain-management mechanism, known as conditioned pain modulation (CPM), in remote body regions.
The upper arm is stimulated to control the migraine pain in the head.
The dual-use indication of the Nerivio device will allow patients to use the wearable more frequently and proactively prevent migraines.
Theranica CEO and co-founder Alon Ironi said: “While there is an established desire for effective non-drug options, especially for adolescents, migraine care needs to treat the whole person given the nature of this long-term disease.
“Nerivio is paving a bold path forward in migraine treatment and prevention for adolescents and adults.
“We are hopeful this expanded dual-use indication will have a tremendous impact on mitigating the burden of migraine symptoms and improving patient quality of life.”
The latest FDA clearance is supported by data obtained from a randomised, double-blind, placebo-controlled study.
In the trial, Nerivio was used every other day and showed a significant reduction in monthly migraine days while reaching other prevention endpoints.



