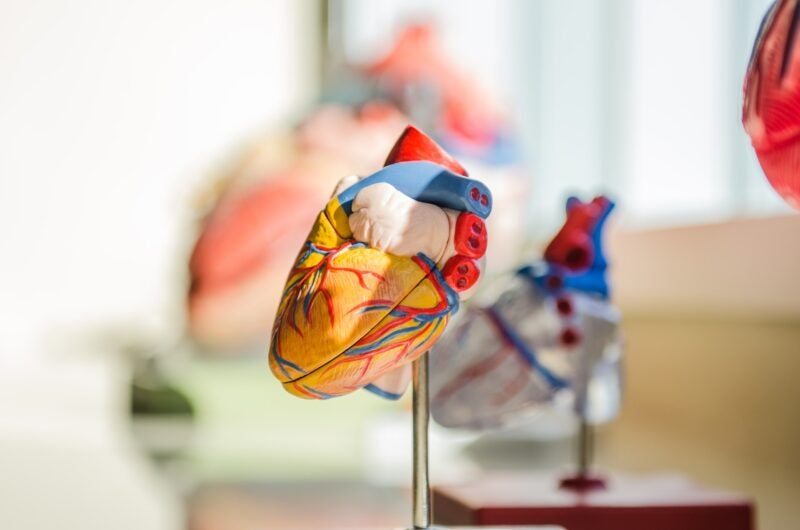
Thubrikar Aortic Valve has concluded the preliminary CE Mark-enabling study of the Optimum Transcatheter Aortic Valve (Optimum TAV) in severe aortic stenosis patients.
The Competent Authority of Poland granted approval for the five-subject study, which was carried out in line with the European Union Medical Device Regulation (EU MDR) and observed by KCRI, a clinical research organisation (CRO).

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
The company noted that better single-digit mean gradients and effective orifice areas (EOAs) were observed compared to the majority of transcatheter aortic valve replacement (TAVR) valves currently available in the market.
Claimed to be the only short-profile, self-expanding TAV in the sector, Optimum TAV offers trouble-free coronary access as well as the advantages of a self-expanding valve.
It is created with dimensional proportionality and leaflet surface geometry that imitates the natural aortic valve, without any suture holes in the leaflet’s flexion zone. It also features a distinctive anti-calcification treatment.
Additionally, the Optimum TAV was demonstrated to have pre-clinical durability of up to 24 years with reduced calcification compared to validated bioprosthetic surgical heart valves.
The value could potentially meet the requirements of a younger patient population, who need a durable valve with exceptional hemodynamic performance.
The first patient in Brazil who had an Optimum TAV implant is doing well at four years.
Thubrikar Aortic Valve president and founder and Optimum TAV inventor Dr Mano Thubrikar said: “We are delighted with these initial results at one-month and six-month outcomes.
“These were truly ‘real world’ patients, including those with horizontal aorta, eccentric or no calcium burden, and small aortic annulus.
“The unique short frame and self-expanding technology of the Optimum TAV performed very well in these complex group of patients, and we look forward to expanding enrolment throughout Europe to obtain CE Mark approval.”





