
Tissue reconstruction company Tissium has secured €50m ($53.8m) in Series D financing round to launch its platform of biomorphic programmable polymers.
The funds came from previous investors Cathay Health, Mutuel Innovation and Sofinnova Partners, as well as fresh investment from Fonds Stratégique des Transitions and Merieux Developpement.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
Based in Paris, France and Boston, USA, the privately-owned company’s pipeline of seven products include sutureless nerve repair, hernia repair and cardiovascular sealants. Each product aims to enhance tissue reconstruction in unique ways.
Christophe Bancel, CEO of Tissium said the round will be used to finance commercialisation of the company’s first products by 2024, as well as extending its product and platform pipeline further.
“This funding significantly bolsters our ability to move with speed towards our goal of enhancing tissue reconstruction for patients. We look forward to continuing our work to bring innovation in the space and develop products that make a difference in patients’ lives,” said Bancel.
Despite operating in a challenging funding environment, Bancel said Tissium’s in-house manufacturing of the technology and the wide range of its potential uses in atraumatic body repair had made it an attractive prospect for investors
As Tissium expands its platform to new therapeutic areas, the company said it will focus on introducing their portfolio to the US market, with expansions into the EU and elsewhere planned further down the line.
This follows recent successes in the ongoing first in-human study on Tissium’s COAPTIUM Connect, combining a 3D-printed implantable device and on-demand activated adhesive to achieve sutureless nerve repair. It is the first time Tissium has been used in a clinical study and will pave the way for future trials in the near term.
Tissium’s 3D printing applications could prove extremely lucrative in the future, as the company hopes to co-develop surgical solutions with strategic partners.
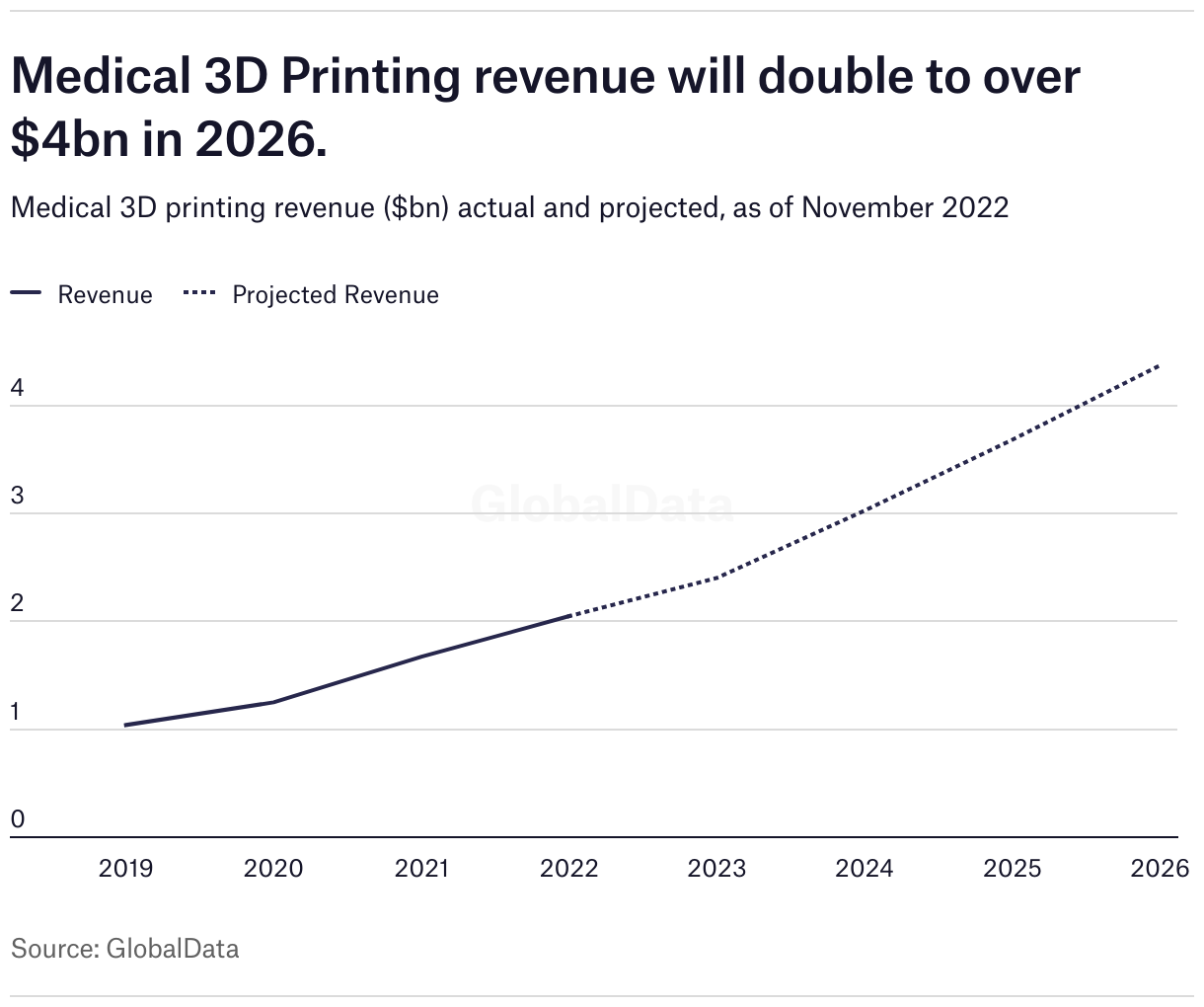
By providing cheaper, easily produced and personalised medical devices, 3D printing is expected to be one of the industry’s most valuable sectors in years to come. According to GlobalData analysis, medical and dental 3D printing revenue has been steadily increasing since 2018 and is set to continue until at least 2026.
The Paris based company has benefitted from France’s strong medtech start-up landscape. Since 2020, Tissium has been a participant in the French Tech 120 program designed to support technology startups. French med tech will also benefit from the $8bn (€7.5bn) Health Innovation Plan 2030, with $2.5bn (€2.4bn) allocated to startups in the sector.





