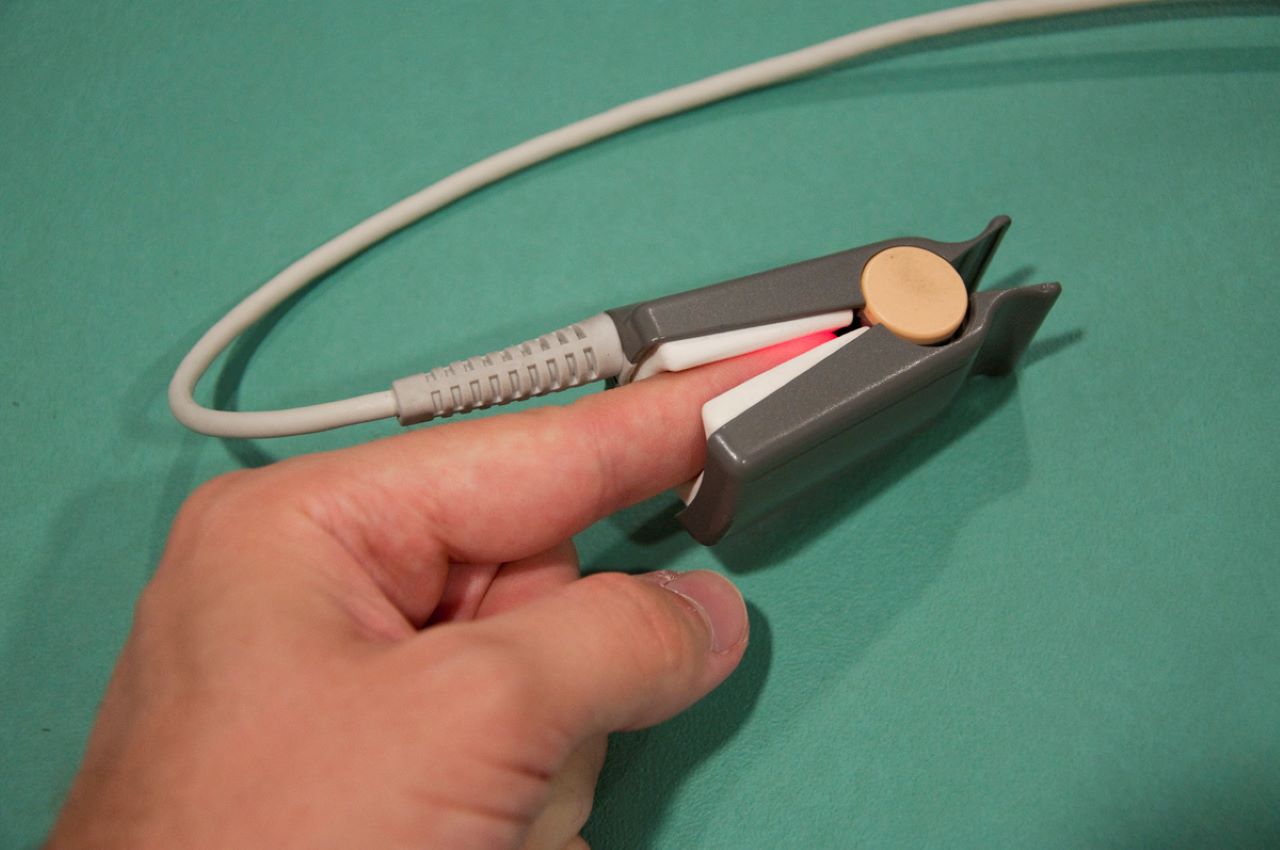
Telehealth company Tyto Care has launched its US Food and Drug Administration (FDA)-cleared fingertip Pulse Oximeter (SpO2) medical device for checking blood oxygen saturation levels and heart rate remotely.
The virtual examination solution used in the home setting can support clinicians in monitoring patients with Covid-19 and chronic lung or heart conditions, helping them avoid hospital and clinic visits.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
The Pulse Oximeter connects to TytoCare device by a cable. Users can carry out blood oxygen exam by placing their finger in the SpO2 device for 15 seconds.
The blood oxygen and heart rate results will soon appear on the handheld device and the Pulse Oximeter adaptor. The results are logged in the TytoCare platform for clinician’s review and are sent to patients’ Electronic Health Record (EHR).
Furthermore, if the exam is conducted in real time during a live telehealth visit, the clinician can see the results in the TytoCare Clinician Dashboard.
The reliability of results can be evaluated by checking the heart rhythm graph generated during the exam.
Tyto Care CEO and co-founder Dedi Gilad said: “We’re excited to announce the release of our Pulse Oximeter, providing patients with more tests that bring the clinic directly to them.
“The Covid-19 pandemic thrust telehealth into the spotlight, and we are constantly enhancing the TytoCare platform to ensure users have access to the most comprehensive telehealth solution available.
“The pandemic will eventually be behind us, yet telehealth will remain a key component in the future of healthcare, providing patients with the best possible remote care and clinicians with actionable insights into their patients’ health.”
Through partnering health systems, Tyto Care’s Pulse Oximeter is available in the US and Israel.
The company plans to make the device available in Europe, South Africa, and Asia in the future.





