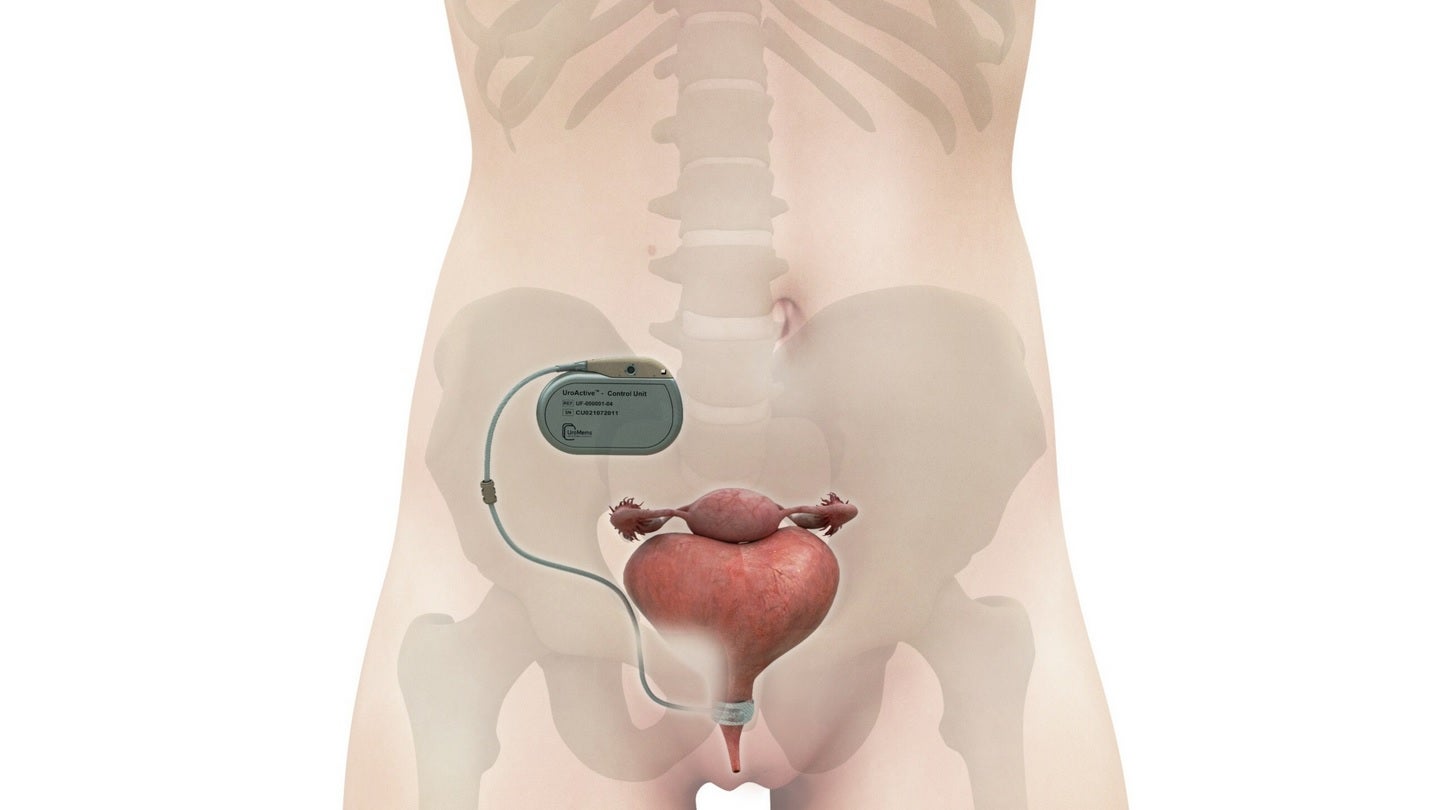
UroMems has announced the first implantation of its smart, automated artificial urinary sphincter (AUS), UroActive, in a female patient as part of a clinical study.
UroActive, which is powered by a MyoElectroMechanical System (MEMS), is claimed to be the first smart active implant designed for the treatment of stress urinary incontinence (SUI).

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
The company will utilise data from this trial for the design and implementation of the pivotal clinical study in the US and Europe.
Professor Emmanuel Chartier-Kastler, Dr Aurélien Beaugerie and Dr Christophe Vaessen conducted the robotic-assisted procedure for the female recipient at La Pitié-Salpêtrière University Hospital in Paris, France.
The National Agency for the Safety of Medicines and Health Products approved the first-in-female procedure.
UroMems CEO and co-founder Hamid Lamraoui said: “This is a unique accomplishment for UroMems and SUI in women.
“We are addressing the observations by so many physicians that there is a vast unmet need for a better way to definitively treat this female population.”
The company has designed the UroActive implantable electronic AUS to treat sphincter insufficiency in both male and female patients with SUI.
In November last year, the company announced the first-in-human (male) UroActive implant.
Recently, the company secured the Safer Technologies Program designation from the US Food and Drug Administration for this implant.
SUI, also known as involuntary urinary leakage, is expected to affect around 90 million Europeans and 40 million Americans.
This condition arises when the pressure within the bladder surpasses that of the muscle (sphincter) surrounding the urethra.





