The US Food and Drug Administration (FDA) has granted 510(k) clearance for Vektor Medical’s new computational electrocardiogram (ECG) mapping system, vMap.
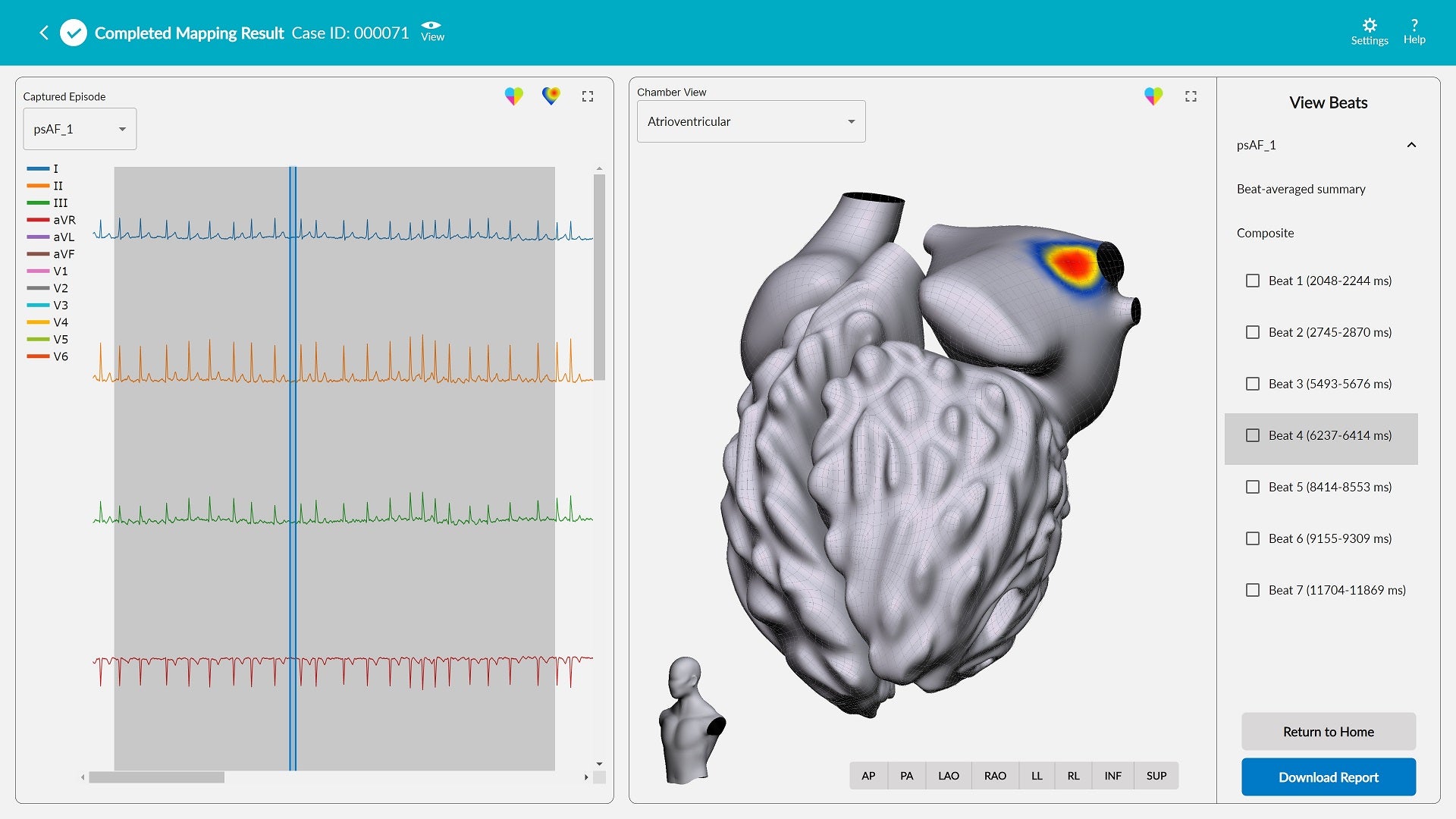
The non-invasive system can be used to map potential arrhythmia sources, referred to as hot spots, linked with stable and unstable arrhythmias anywhere in the heart using ECG data. It is capable of creating actionable 2D and 3D maps of potential arrhythmia sources using computational modelling.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
The FDA clearance will enable Vektor Medical to commercially market the vMap technology to users across the US.
According to Vektor Medical, the easy-to-use system has succeeded in detecting arrhythmia sources for a wide range of arrhythmias, including atrial fibrillation.
Clinicians can input case information, download and mark up an ECG in the system, and then generate a three-dimensional, interactive arrhythmia hot spot map visualising both the inside and outside of the heart, all in less than three minutes.
Vektor Medical CEO Mike Monko said: “Cardiac arrhythmias impact millions of people across the globe, increasing the risk of serious health-related issues, such as stroke, heart failure and even death. Yet, today’s therapies have significant issues – drug therapy can have severe side effects and non-targeted ablation has disappointingly low success rates.
“With vMap we are changing how electrophysiologists think about mapping. By providing a hot spot map in only minutes based on non-invasive ECG data, physicians can create a more effective ablation plan and spend less time finding target locations.
“Our goal is to increase first-pass success rates, lower risk and decrease the current cost burden of ablation on the healthcare system.”
In planning and procedural settings, vMap can also be used as a non-invasive standalone tool or to complement traditional invasive electro-anatomical mapping systems.





