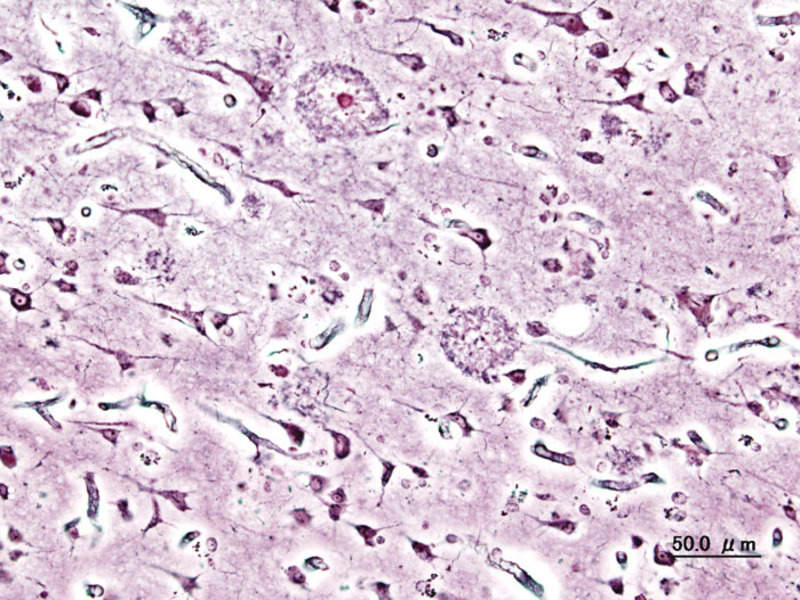
The US Food and Drug Administration (FDA) has granted Breakthrough Device designation to digital therapeutic company Dthera Sciences for its development-stage product DTHR-ALZ.
If approved, DTHR-ALZ would become the first non-pharmacological prescription treatment for Alzheimer’s disease.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
Dthera Sciences Scientific and Regulatory Affairs vice-president Dr Martin Culjat said: “This Breakthrough designation has provided us with a remarkable opportunity to expedite the development of our digital therapeutic device.
“We feel deeply honoured that the FDA has confirmed DTHR-ALZ meets the requirements for this designation, and we are eager to work together with the FDA throughout this process.”
The Dthera Sciences product can be used to reduce the symptoms of agitation and depression associated with a major neurocognitive disorder of the Alzheimer’s type.
Dthera Sciences chief executive officer Edward Cox said: “Alzheimer’s disease is a significant and rapidly growing public health crisis, and, among the ten leading causes of death in the US, it is the only one that cannot be prevented, cured or slowed.
“We commend the FDA for recognising this significant unmet medical need as well as the critical importance of providing innovative new treatments to patients with Alzheimer’s and their caregivers.”
The DTHR-ALZ device has been designed to potentially serve as a prescription digital therapeutic that will deliver Reminiscence Therapy to patients with Alzheimer’s disease.
Reminiscence Therapy is an evidence-based psychosocial intervention which, as shown in clinical trials, can improve symptoms of the disorder.
The device will use artificial intelligence to automatically optimise the therapy based on various forms of biofeedback from the patient, and will provide Reminiscence Therapy with more frequency, consistency and personalisation while requiring minimal time and resources.





