The US Department of Health and Human Services (HHS) has unveiled a pilot programme with five states to use portable, cartridge-based Covid-19 molecular test kits from Cue Health that provide rapid results.
The programme will analyse how to best integrate Cue Health’s diagnostic technology into disease surveillance policies and infection control in institutions such as nursing homes.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
It is part of a $481m contract awarded last month by HHS in collaboration with the Department of Defense (DoD) to Cue Health.
The contract was awarded to expand production capacity for the test in the US.
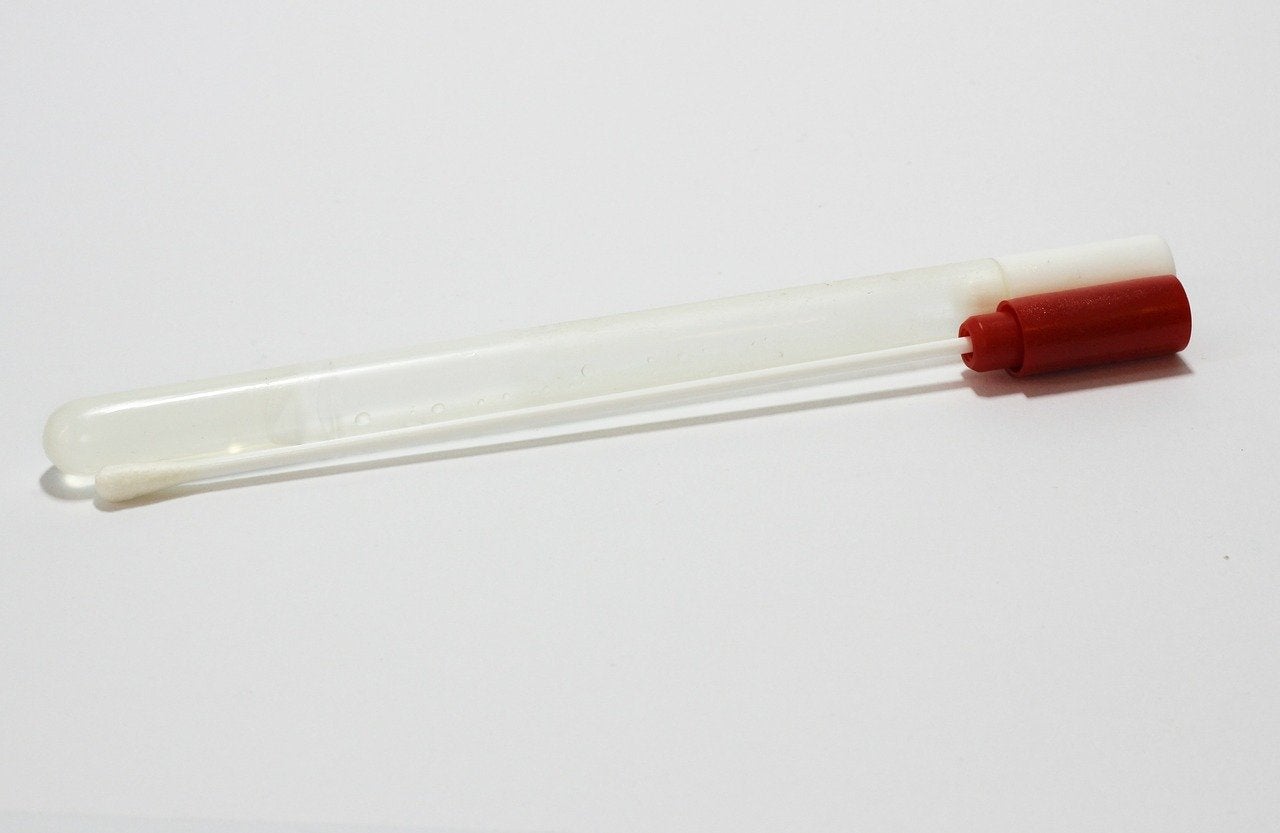
Used by the National Basketball Association bubble and healthcare providers in the country, the nasal swab primary molecular point-of-care (POC) test helps to limit the spread of Covid-19.
It detects the virus’s ribonucleic acid (RNA) or genetic material and provides results in about 20 minutes.
HHS distributed a total of 27,000 test kits, which include the Cue Sample Wand (nasal swabs), Cue Covid-19 Test Cartridges and 600 Cue Health Monitoring Systems (Cartridge Readers).
Testing will be carried out by a trained health professional who takes a sample from the lower part of the nose using the wand.
The wand is then inserted into the cartridge, which is coupled to the Cue Health Monitoring System. After the cartridge reader evaluates the specimen, it transmits the results to the Cue Health App on a connected mobile smart device.
HHS Health ADM assistant secretary Brett Giroir said: “The Cue Health testing system will undoubtedly be a valuable addition to our testing ecosystem.
“Having a highly specific and sensitive POC molecular test could dramatically improve infection control at nursing homes and other institutional settings especially.”





