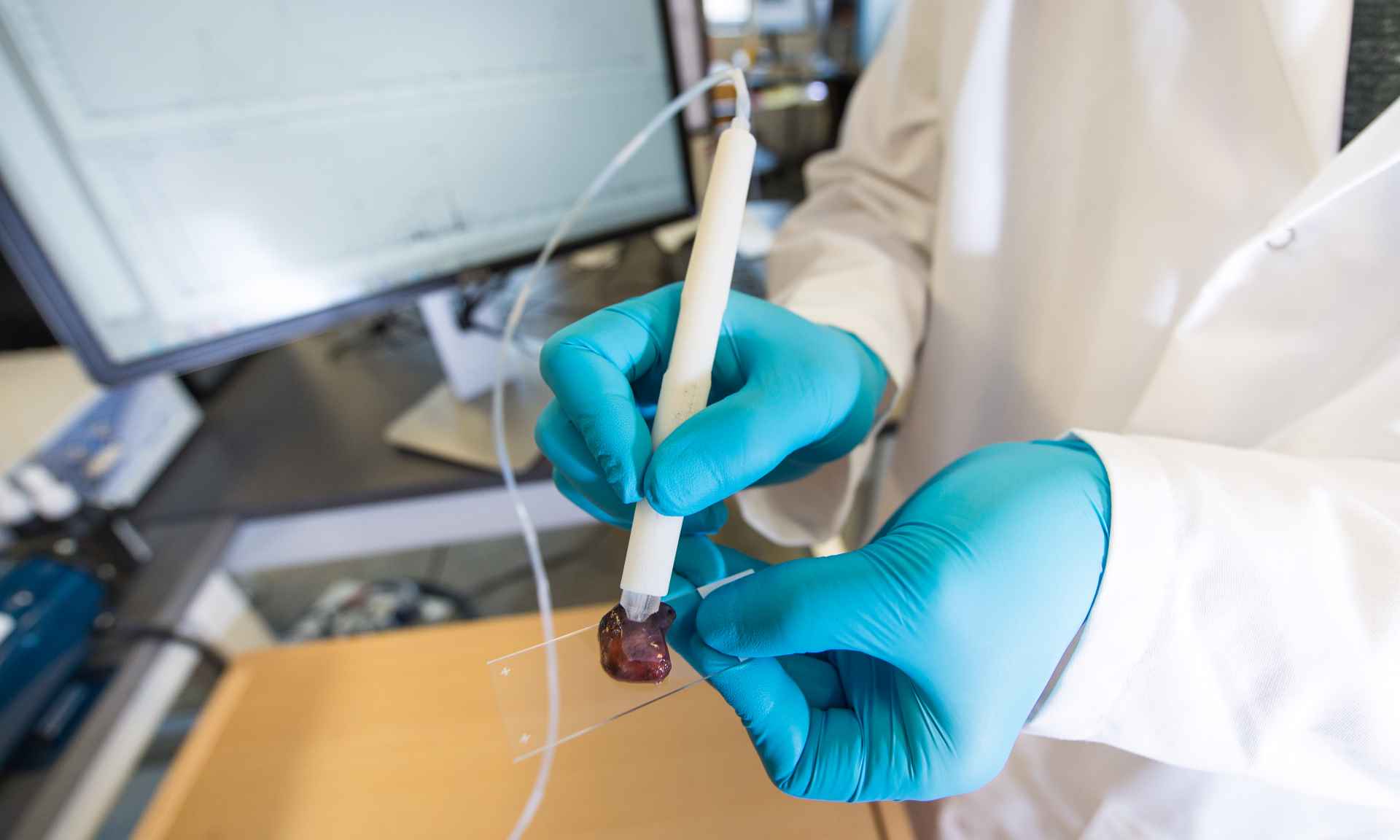
The University of Texas at Austin (UT Austin), US, researchers have tested a diagnostic device, named MasSpec Pen, in pancreatic cancer surgery.
A handheld tool, the device was able to precisely detect tissues and surgical margins directly in patients as well as distinguish between healthy and cancerous tissue from banked pancreas samples.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
Compared to the existing gold standard diagnostic, Frozen Section Analysis, the latest method is approximately 100 times quicker at about 15 seconds per analysis.
The capability to precisely detect margins between healthy and cancerous tissue in pancreatic cancer surgeries can increase survival chances for patients, the researchers said.
MasSpec Pen was developed by the UT Austin team in alliance with Baylor College of Medicine head of endocrine surgery, James Suliburk.
In the latest study, investigators initially used the MasSpec Pen to assess 157 banked human pancreatic tissues to develop and analyse the technology in the lab for pancreatic cancer.
Afterward, the system was shifted to the operating room at Baylor College of Medicine’s Baylor St Luke’s Medical Center in Houston. At the facility, the device was used by surgeons in 18 pancreatic surgical procedures.
So far, the pen has been analysed in more than 150 human surgeries, including breast and thyroid procedures.
UT Austin chemistry assistant professor Livia Schiavinato Eberlin said: “These results show the technology works in the clinic for surgical guidance.
“Surgeons can easily integrate the MasSpec Pen into their workflow, and the initial data really supports the diagnostic accuracy we were expecting to achieve.”
Cancer surgeons face difficulties in detecting the margin between healthy and cancerous tissue during surgery.
If cancerous tissue is not removed, there exists a risk of tumour regrowth. Meanwhile, excessive removal of healthy tissue, particularly from vital organs, can impact the health of a cancer patient.
MasSpec Pen is expected to address these challenges.
The research team intends to seek US Food and Drug Administration approval for MasSpec Pen as a medical device.





