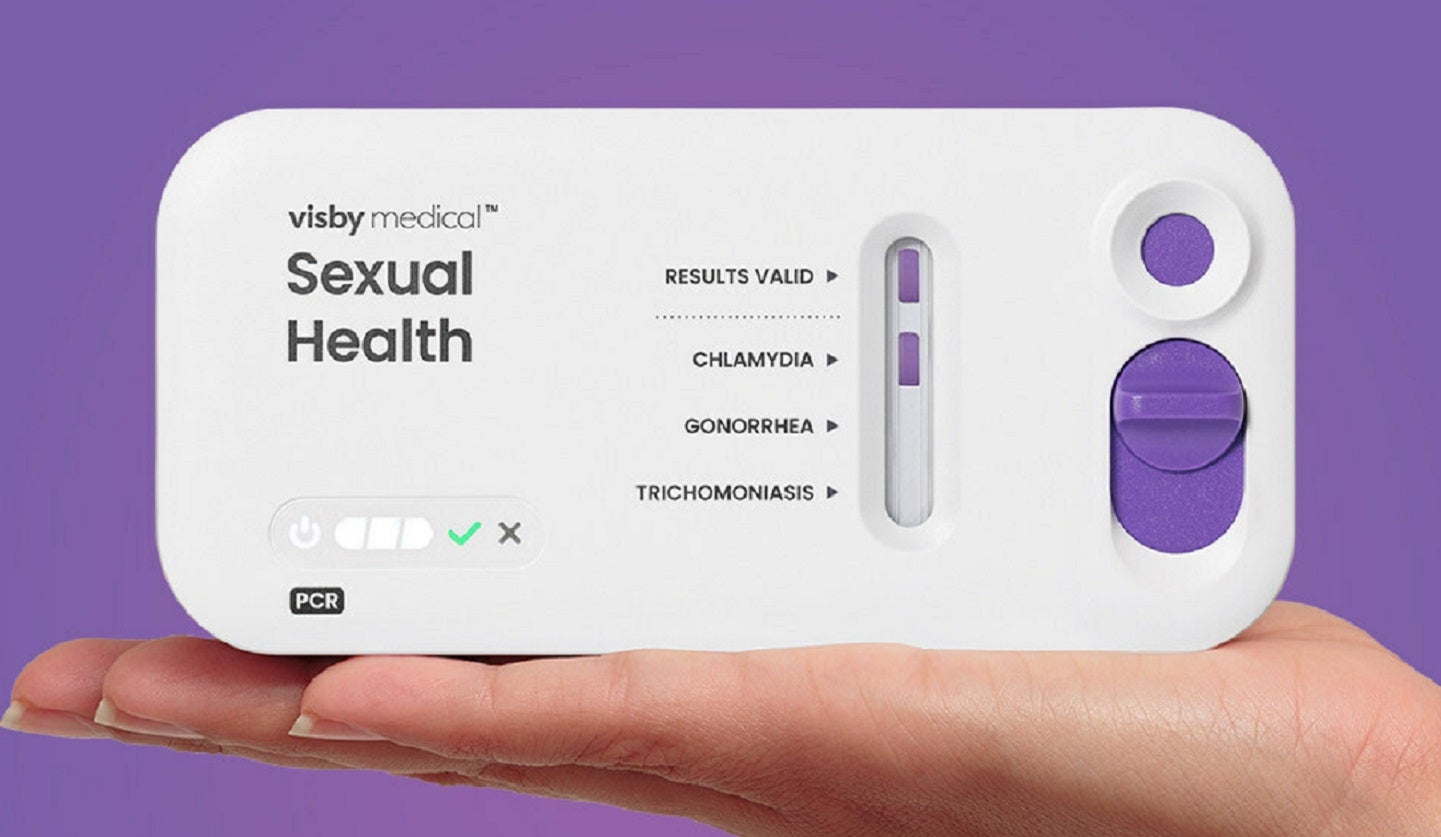
Visby Medical has received 510(k) clearance from the US Food and Drug Administration (FDA) for its second-generation Sexual Health Test.
The Sexual Health Test is a fast polymerase chain reaction (PCR) diagnostic test that aims to uncover sexually transmitted infections (STIs) in women caused by Chlamydia trachomatis, Neisseria gonorrhoeae and Trichomonas vaginalis.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
Visby also secured a Clinical Laboratory Improvement Amendments (CLIA) waiver from the FDA for the point of care (POC) test.
Said to be an ‘instrument-free’ platform that fits in the palm of the user’s hand, the CLIA-waived, Sexual Health test provides results in less than 30 minutes for clinicians in POC settings.
Visby Medical Operations senior vice-president Mark Medlen said: “We are excited that the clearance of the second generation Sexual Health Test will enable us to move production to our fully automated lines, allowing us to deliver more tests to eagerly waiting customers.”
Featuring a sleeker design, the new single-use, fast PCR platform is said to offer significant improvements in workflow and reliability compared to its predecessor.
Henderson, Nevada, CareNow Urgent Care market medical director Lamont Tyler said: “Visby innovation has become essential for maintaining quality urgent care and positive patient experience in our practice.
“The Visby test is a win-win, helping fight antibiotic resistance as well as the spread of untreated infection while empowering patients to engage health services with greater confidence.”
In January, the US FDA granted Visby Medical emergency use authorization (EUA) for its new Respiratory Health POC test, a rapid PCR device, for use in CLIA-waived settings.





