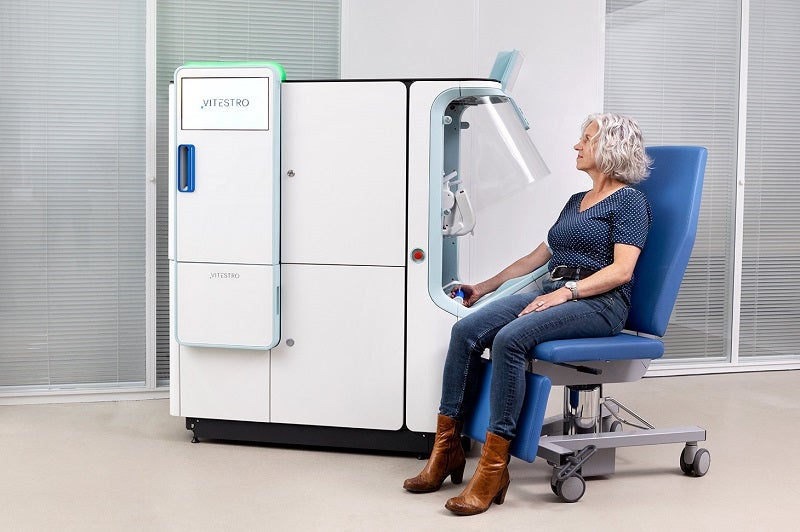
Dutch medical robotics company Vitestro has revealed an advanced autonomous blood drawing device.
The new device combines artificial intelligence (AI) and ultrasound-guided 3D reconstruction with robotic needle insertion to ensure the precise and secure collection of blood.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
The blood drawing device is intuitive to use and empowers patients to be self-efficacious during the complete blood collection procedure.
It allows almost full automation of the pre-analytical phase of the process.
The laboratory automation technology has the potential to reduce high blood test error rates, which are mainly caused by manual variability.
Vitestro said that it has already performed 1,500 automated blood draws in more than 1,000 patients with its prototype in clinical studies.
Vitestro co-founder and CEO Toon Overbeeke said: “The mounting shortage of healthcare personnel is imminent.
“The pandemic has further led to a loss of workforce, causing a bleak outlook for hospital output around the globe, and leading to reduced access and continuity of care.
“That’s why revolutionary automation like our blood drawing device is inevitable to solve the industry’s biggest problem.”
Initially, the company plans to implement the new device in outpatient phlebotomy departments, where patients can choose the new venipunture device or standard methods.
The technology can be used by patients aged 16 years and above, as well as those with comorbidities or puncture difficulty.
Vitestro plans to begin a pivotal clinical trial of the new device for regulatory approval in Europe from 2023 and expects to bring it to the European market in 2024.
According to the company, blood collection, known as phlebotomy, is the most common invasive medical procedure.
The automation of labour-intensive procedures can help to transform the quality and availability of care.



