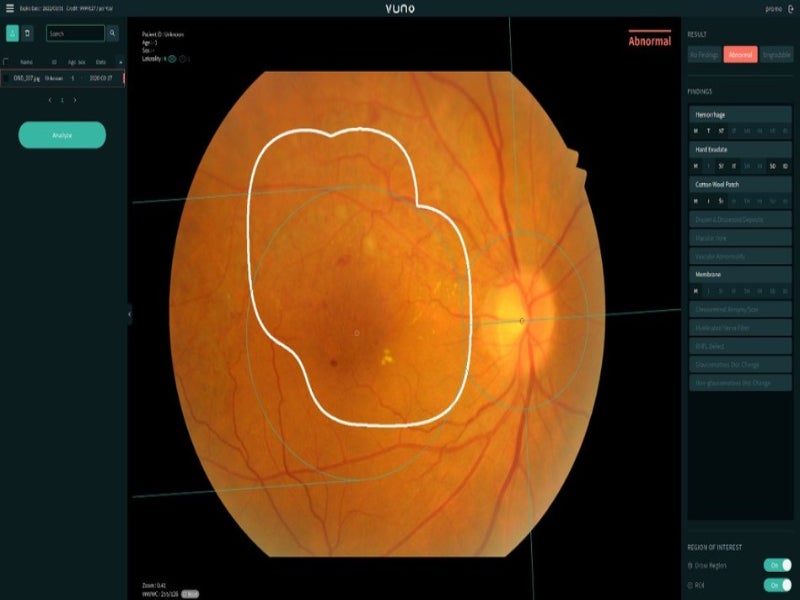
VUNO, a Born2Global Centre member company, has secured Class III medical device approval from the Korean Ministry of Food and Drug Safety for its VUNO Med-Fundus artificial intelligence (AI) screening solution.
Clinically proven to deliver highly accurate screening results, VUNO Med-Fundus AI is considered to be the first-ever AI device to gain a Class III approval in Korea.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
By analysing retinal fundus images, the device can detect and locate more than 12 lesions within one second. The findings detected include haemorrhage, hard exudate, chorioretinal atrophy or scar, vascular abnormality, retinal nerve fibre layer defect and glaucomatous disc change.
Additionally, it encompasses all findings that are important in diagnosing retinal diseases, including diabetic retinopathy, macular degeneration and glaucoma.
The examination is performed to observe the state of the fundus that plays a significant role in eyesight. Fundus is made up of the retina, optic nerve and retinal blood vessels.
This procedure helps in the early detection of diseases such as diabetic retinopathy, macular degeneration and glaucoma that may lead to loss of sight.
VUNO Med-Fundus AI was developed based on a large-scale deep learning dataset of more than 100,000 fundus images.
More than 50 ophthalmologists, including 28 specialists, examined the images.
An external validation exam, incorporating multi-ethnic, multi-centre settings, verified a stable performance by the solution matching that of retina specialists.
A research paper published in Ophthalmology revealed that the solution’s clinical validation (AUROC) of the 12 findings resulted in an impressive range of 96.2% to 99.9%, and 94.7% to 98.0% with external datasets.





