Zeus Scientific has received US Food and Drug Administration (FDA) clearance for new indications of four of its diagnostic tests intended for Lyme disease.
The assays are the Zeus ELISA Borrelia VlsE1/pepC10 IgG/IgM, Borrelia burgdorferi IgG/IgM, Borrelia burgdorferi IgM and Borrelia burgdorferi IgG Test Systems.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
The tests involve concurrent use of two enzyme immunoassays (EIA), instead of the existing two-step process requiring an initial EIA test followed by a protein test called Western Blot.
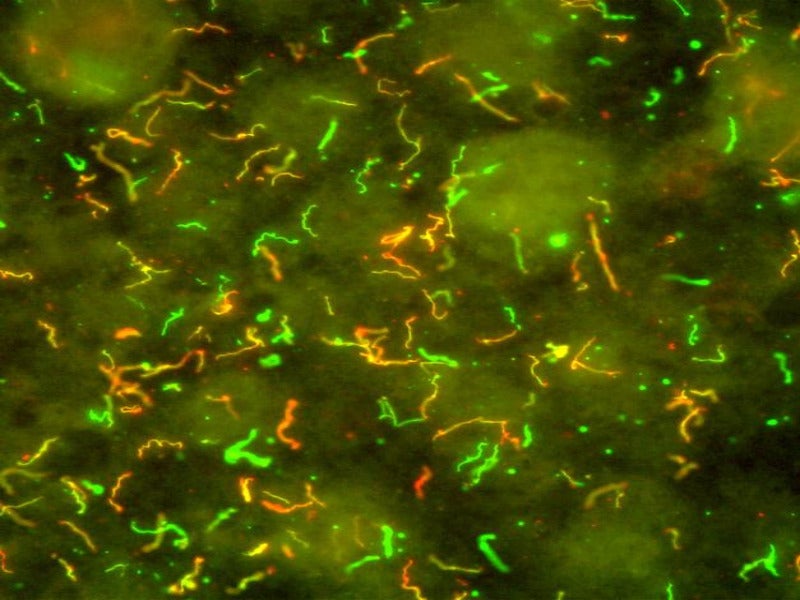
Lyme disease tests are designed to analyse a blood sample for antibodies against Borrelia burgdorferi, the causative bacteria which is transmitted via infected ticks.
The disease is characterised by fever, headache, fatigue and skin rash. It could spread to joints, the heart and nervous system.
FDA Office of In Vitro Diagnostics and Radiological Health director Tim Stenzel said: “Lyme disease can have a devastating impact on patients. With today’s action, clinicians have a new option to test for Lyme that is easier to interpret by a clinical laboratory due to the streamlined method of conducting the test.
“These tests may improve confidence in diagnosing a patient for a condition that requires the earliest possible treatment to ensure the best outcome for patients.”
The FDA clearance is based on results from clinical studies, where the accuracy of the new approach leveraged by Zeus Scientific assays was found to be similar to that of existing methods in identifying antibodies to Borrelia burgdorferi.
The company has also announced the launch of its FDA-cleared Zeus Borrelia Modified Two-Tiered Testing (MTTT) algorithm.
The algorithm leverages the Zeus ELISA Borrelia VlsE1/pepC10 IgG/IgM Test System as a first-tier test, followed by testing of positive or equivocal specimens using a second Zeus ELISA Test System.





