Alafair Biosciences has received 510(k) clearance from the US Food and Drug Administration (FDA) for its VersaWrap class II medical device.
The approval enables application of the bioresorbable hydrogel implant in all patient populations, irrespective of their age.
It allows paediatric surgeons and medical facilities to treat younger patients using the device.
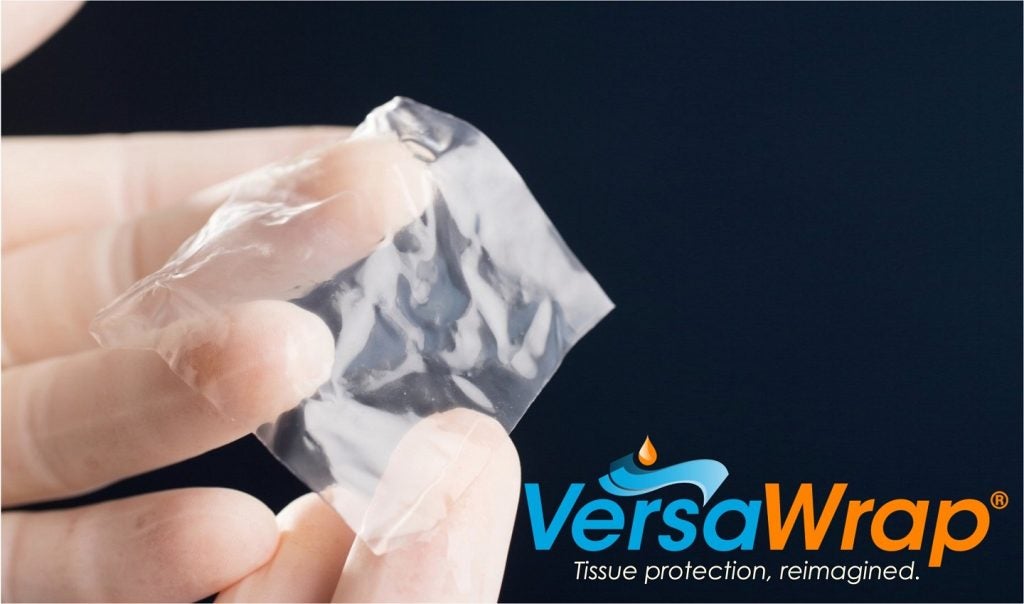
VersaWrap creates a gelatinous coating around target tissues such as tendons, peripheral nerves and surrounding soft tissues.
This reduces postoperative tethering and facilitates gliding during healing, providing consistent outcomes.
It is primarily utilised in a range of tendon and peripheral nerve treatments by orthopaedic, reconstructive plastic and spinal surgeons. It can be implanted as a gel or sheet.
Alafair chief scientific officer Sarah Mayes said: “VersaWrap is designed to be gentle and intuitive to improve health outcomes for all ages, including the youngest and most vulnerable populations.
“VersaWrap is simple to implant as it can be tailored to the size, geometry, and location of target tissues. Specifically, the gel version is designed to meet the needs of anatomically challenging injuries, making the device attractive for use in smaller patients."
The new clearance opens additional markets for the company as it expands VersaWrap’s use in surgical injuries repair in younger populations, including fractures or congenital abnormalities, accidents, and sports-related activities.
Cook Children's Healthcare System hand surgery medical director Pamela Sherman said: “Post-operative scarring is prevalent in all ages. Scarring is compounded in children as early motion is often a challenge in this incredibly active patient population.
“The ability to use VersaWrap to avoid adhesions following complex repairs in the hands of young patients brings confidence that they can recover without the limitations associated with soft tissue tethering.”