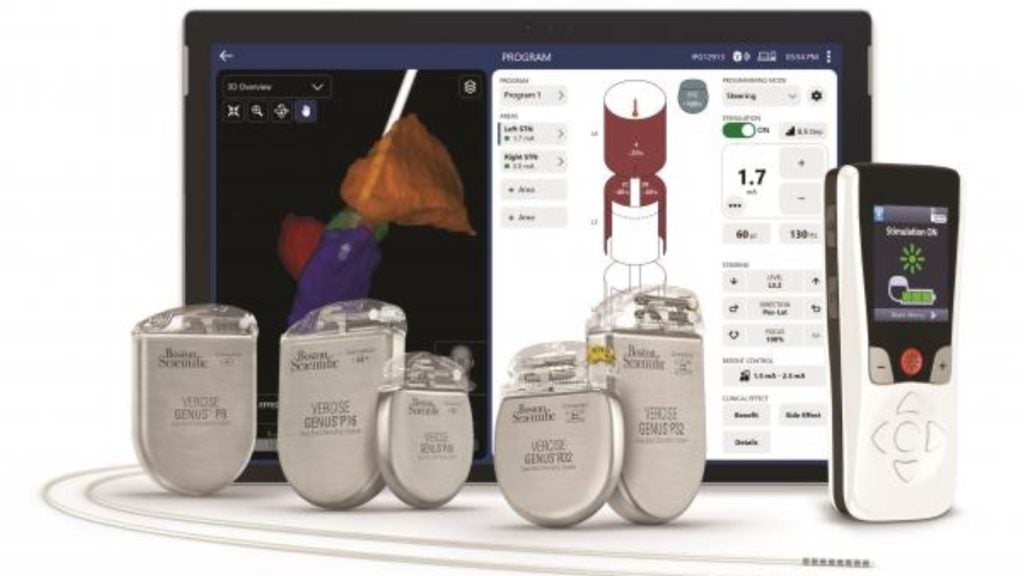
The US Food and Drug Administration (FDA) has granted approval for Boston Scientific’s Vercise Neural Navigator 5 Software for deep brain stimulation (DBS).
Utilised as a component of the Vercise Genus DBS systems, the software provides clinicians with actionable data for efficiently programming treatment for individuals with essential tremor or Parkinson's disease.
The fourth-generation Vercise Genus DBS system helps deliver targeted electrical stimulation through leads that are surgically implanted in the brain and connected to implantable pulse generators for treating Parkinson’s disease and essential tremor symptoms.
Featuring STIMVIEW XT technology, the new software is the newest inclusion in the company's integrated portfolio of image-guided programming solutions for its DBS system.
Created in collaboration with Brainlab, these tools have shown a 56% reduction in programming time and deliver real-time visualisation and stimulation of the brain anatomy of each person.
The enhanced user interface of the Vercise Neural Navigator 5 Software showcases data related to a patient in a simplified format and helps clinicians access advanced settings for improved delivery of therapy.
Furthermore, the software offers flexibility for better management of increasing requirements of individual patients at any stage of their condition.
Boston Scientific Neuromodulation president Jim Cassidy said: “Developing meaningful tools to help physicians provide personalised treatments for their patients delivers on our promise to advance our technologies for people living with neurological conditions.
“Providing effective DBS therapy is complex and can be time-consuming. This software will help streamline the process and allow for more doctor-patient interaction time.”
In February this year, Boston Scientific secured 510(k) clearance from the FDA for its LithoVue Elite Single-Use Digital Flexible Ureteroscope System.