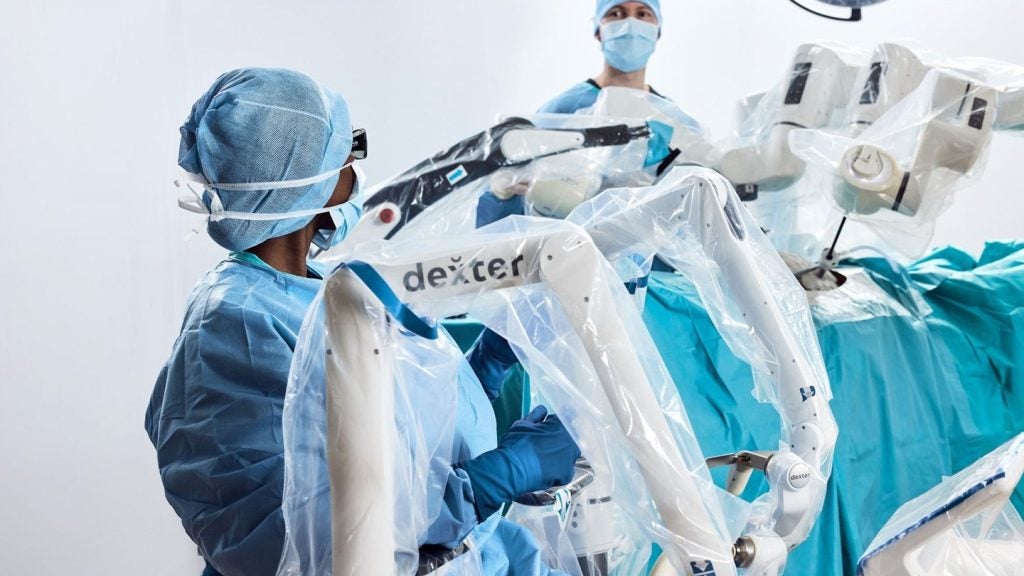
Distalmotion has submitted a 510(k) application to the US Food and Drug Administration (FDA) seeking clearance to use the DEXTER robotic surgery system for sacrocolpopexy, sacrocervicopexy and endometriosis resection.
The submission comes after completing the Robotic Sacrocolpopexy and Sacrocervicopexy trial, known as SPARO, which assessed DEXTER in those procedures.
If the agency clears the submission, it would become the fourth overall and second gynaecology indication for DEXTER in the US, supporting broader service line development in ambulatory surgery centres (ASCs).
Distalmotion said that procedures are continuing to move from hospitals to outpatient settings, reflecting physician, payor and patient preferences. In that shift, robotics is being used to support migration.
For ASCs developing gynaecology programmes, broader robotic indications can enable hysterectomy, sacrocolpopexy and other benign gynaecologic procedures within the same operating day.
The FDA has authorised the company to begin a US myomectomy trial through an investigational device exemption (IDE), supporting further progress in its gynaecology clinical programme.
Distalmotion CEO Greg Roche said: “Expanding gynaecology capabilities in outpatient environments represents an important next step in the evolution of the DEXTER robotic programme.
"DEXTER is designed to help outpatient centres introduce robotics in a practical way, enabling care teams to build sustainable service lines across multiple specialities.”
DEXTER is a soft-tissue surgical system with single-use wristed instruments designed to support precision and dexterity.
Its mobile design fits operating rooms without modification while a sterile console and open architecture support flexible workflows and technology compatibility.
In November 2025, Distalmotion closed a Series G funding round totalling $150m to expedite the US expansion of DEXTER.