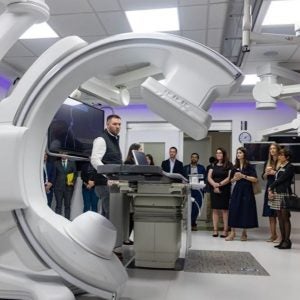
Novo Nordisk Insulin Pen Simulation
Novo Nordisk, a world leader in medical device design and manufacturing, needed deeper understanding of the behaviour of polymer materials undergoing snap-fit loads during both manufacturing and patient use of insulin-delivery pens.
Solution
SIMULIA’s Polymer Customer Review team worked closely with Novo Nordisk to develop advanced software methodology based on Abaqus FEA that helps capture the complex changes that can occur in polymers and affect product integrity.
Benefits
Increasingly refined analyses of material parameters enables Novo Nordisk to design more efficiently and ensure the highest levels of product performance.
Many medical conditions can be treated with tablets, but others require injections under the skin in order for therapeutic drugs to reach the bloodstream. In the case of insulin administration for diabetes treatment, patients need to self-inject the drug daily.
An insulin pen may be small, but it is a precision instrument with a number of complex parts that must work in perfect tandem. Some pens are durable, containing a replaceable drug cartridge, while other disposable ones come pre-fi lled with the drug.
Injection typically involves twisting a short needle onto the pen, turning a dial to the required dose, and pushing a button to deliver the medication under the skin. After a given number of doses are injected, the cartridge is exchanged for a new one (with a durable device) or discarded (with disposable pens).
Download the below document to read more.