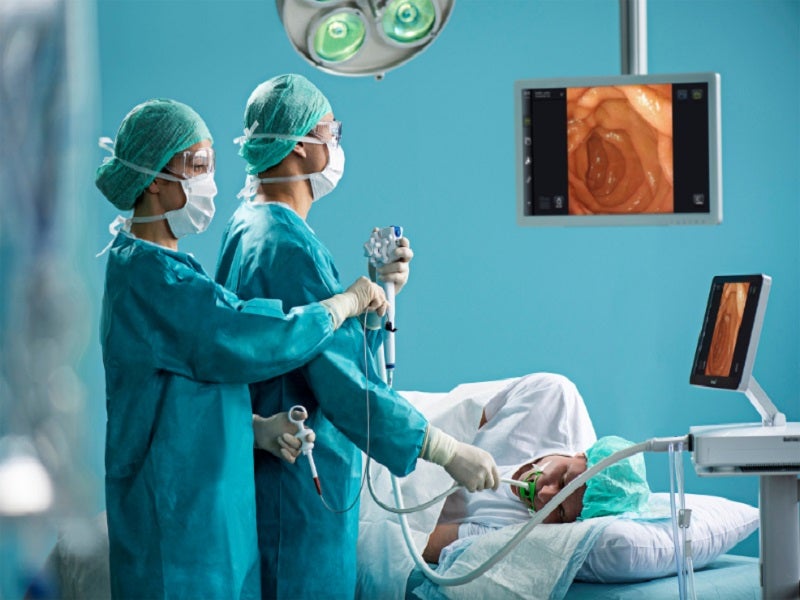
Ambu® aScope™ Gastro is the first sterile single-use gastroscope, developed by Danish medical equipment manufacturer, Ambu. The aScope gastroscope comes with advanced imaging and design features and can be used for a variety of endoscopic procedures.
The single-use aScope Gastro solution, combined with the company’s next-generation displaying and processing unit Ambu® aBox™ 2, will help hospitals to perform quality and cost-effective gastroscopies at ease, across a wide range of care settings including endoscopy units, operating rooms (OR), intensive care units (ICU), emergency rooms (ER), and Ambulatory surgery centres (ASC).
Hospitals using it will benefit from having a single-use scope with a small footprint and less up-front capital investment than a reusable setup.
Ambu entered the gastroscopy market with the launch of the aScope Gastro. In this sector, approximately 20 million procedures a year are being performed with reusable endoscope devices.
Ambu received 510(k) regulatory clearance for Ambu® aScope™ Gastro, and Ambu® aBox™ 2, from the US Food and Drug Administration (FDA) in February 2022.
aScope Gastro features and specifications
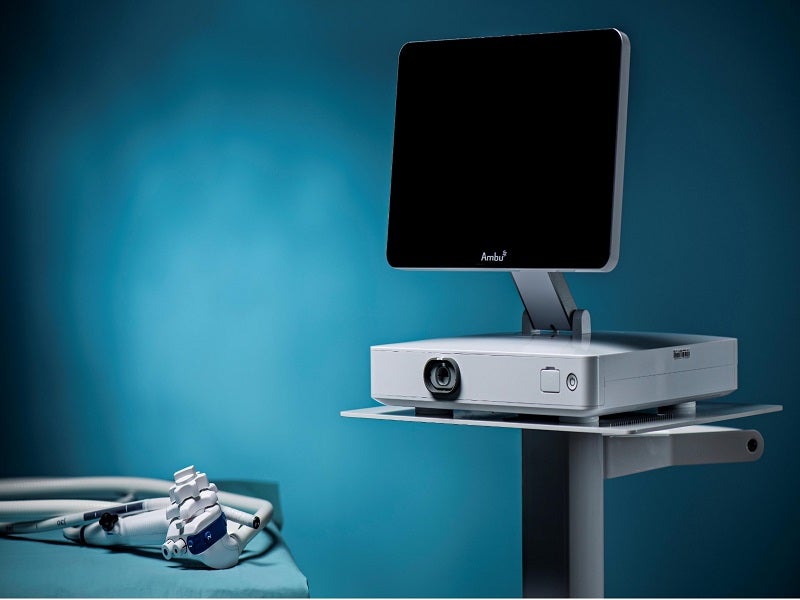
The aScope Gastro single-use gastroscope can be simply unpacked and attached to the Ambu® aBox™ 2 display and processing device, with a built-in touchscreen HD LCD monitor.
The aBox™ 2, with live endoscopic HD capabilities, will provide excellent image quality and is central to the Ambu’s endoscopic ecosystem.
The intuitive user interface is placed next to the endoscopic image on the screen, making it easy to retain focus and control. The touchscreen display can be used as the primary or secondary monitor. It can be adjusted according to different demands and room configurations.
The aScope Gastro has four endoscope buttons, to programme up to eight easily configurable functions; a lightweight handle, with a rippled surface design; and a working channel, with a 2.8mm inner diameter.
The gastroscope has a lightweight connector that is compatible with standard accessory devices and tube sets. The scope has a retroflection angle of 210º and a working length of 1030mm. Its distal tip features dual LEDs providing a 140º field of clear view of anatomical structures.
The combination of the Ambu® aScope™ Gastro sterile single-use gastroscope and the Ambu® aBox™ 2 portable display unit, with advanced imaging and design capabilities, will allow for the expansion of endoscopy into new settings.
Benefits of aScope Gastro
The aScope Gastro module offers portability, advanced technology, and enhanced performance for gastroscopy operations, with low capital investment and zero hidden expenses.
As per Ambu, the single-use aScope Gastro will help healthcare organisations overcome personnel shortages and reduce waiting lists.
The Ambu® aScope™ Gastro solution will streamline the workflow by reducing reprocessing and a number of complicated steps required in the conventional reusable endoscopies.
Moreover, the reusable endoscopes are used numerous times a day and are exposed to severe reprocessing, which might have an impact on performance.
In comparison, the single-use gastroscope ensures consistent quality, without involving such deterioration in mechanical performance.
It also eliminates the possibility of cross-contamination for vulnerable patients.
Marketing commentary on Ambu
Established in 1937, Ambu is a medical device company headquartered in Ballerup, near Copenhagen, Denmark. The company currently employs nearly 4,500 employees in Europe, North America, and the Asia Pacific.
Ambu is the market leader in single-use endoscopes. It sold more than 1.5 million single-use endoscopes in 2021, which was more than that of all other single-use endoscopy companies combined.
The company provides the most comprehensive collection of single-use endoscopic instruments, including disposable endoscopes, and high-performance display units for pulmonology, otorhinolaryngology, gastroenterology and urology. The range will be further expanded with the launch of 20 additional single-use endoscopy devices by 2022 to 2023.