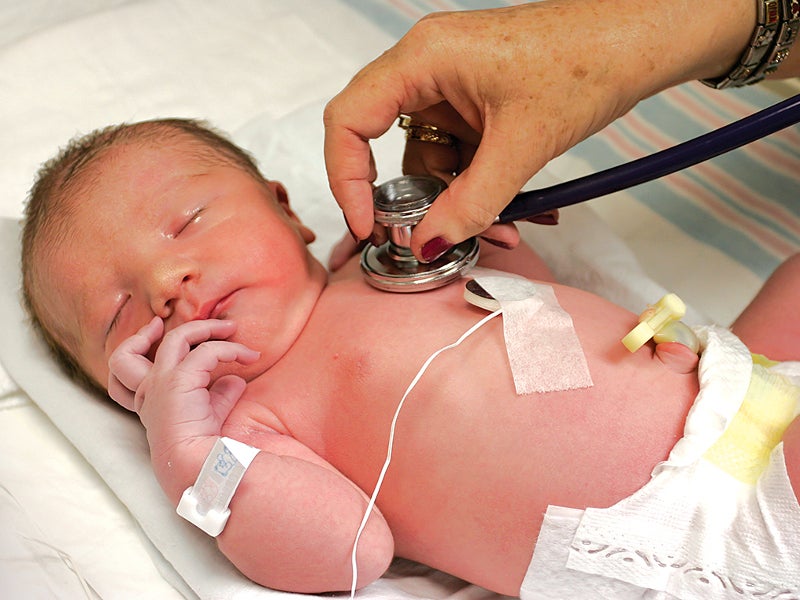
The Amplatzer Piccolo™ Occluder is a self-expanding, wire mesh device approved for the treatment of patent ductus arteriosus (PDA), a common congenital disorder that results in a life-threatening opening in the heart of premature infants and newborns.
Developed by Abbott, the device received 510(k) certification from the US Food and Drug Administration (FDA) for clinical use in January 2019. The device was first implanted in a premature baby with PDA at the Le Bonheur Children’s Hospital in March 2019.
The system received the Conformité Européenne (CE Mark) in Europe in September 2019 and in Australia in March 2020. It was subsequently launched in India in June 2021.
In December 2025, Abbott obtained FDA clearance and the CE Mark for the Amplatzer Piccol Delivery System for use with the Amplatzer Piccolo Occluder. The new delivery system has been engineered to treat PDA in preterm infants, including those weighing as little as two pounds (900g).
Amplatzer Piccolo Occluder design and features
The Amplatzer Piccolo Occluder is a pea-sized, minimally invasive, self-expanding, percutaneous trans-catheter occlusion device used for non-surgical closure of PDA in patients. It is a wire mesh unit made using nitinol and interwoven polyester for remote promotion of tissue growth.
The device is deployed via an anterograde (venous) or a retrograde (arterial) approach. It features a 3mm–5mm-diameter central waist and two retention discs with a diameter of 4mm–6.5mm. The length between the two retention discs ranges from 2mm to 6mm.
The central waist allows for proper positioning of the device in the ductus, while the retention discs are placed either at the ends of the pulmonary and aorta of the ductus or completely inside the duct in very small infants. The discs offer secure positioning in the blood vessels and decrease embolisation.
The occluder is equipped with radiopaque marker bands at each end, which offer visibility during the fluoroscopy process. Other components of the device include a hoop dispenser, a delivery wire, an occluder protector tube and a micro screw attachment.
Amplatzer Piccolo Delivery System details
The Amplatzer Piccolo Delivery System is intended for the placement of the Amplatzer Piccolo Occluder within a PDA.
The system includes a delivery catheter, a loader, a Tuohy-Borst haemostasis valve with extension tube and stopcock, and a self-sealing haemostasis valve. It is supplied in a 4 French size with a usable length of 45cm.
The delivery catheter acts as the conduit for delivering the occluder to the heart. Its distal end incorporates a curve designed to support co-axial positioning of the delivery catheter within the PDA. A curve indicator on the hub provides an additional visual reference for the orientation of this curvature.
The Tuohy-Borst haemostasis valve with extension tube and stopcock enables flushing of the delivery catheter and helps manage blood backflow.
The loader is used to introduce the occluder into the delivery catheter, while the self-sealing haemostasis valve permits flushing of both the loader and the delivery catheter and also helps control blood backflow.
The new delivery system is designed to streamline the implant procedure by using a single catheter rather than multiple catheters. Its shorter, softer configuration is intended to support more precise device positioning in very small infants and newborns.
Functioning mechanism of Amplatzer Piccolo Occluder
The Amplatzer Piccolo Occluder is prepared by threading the occluder on the delivery wire, which is also fitted with a loader and a self-sealing haemostasis valve. It is then introduced into the patient’s body over a guidewire through a small incision made in the patient’s leg and is navigated through the blood vessels into the patient’s heart, where it seals the opening in the heart.
The Amplatzer Piccolo Occluder can be easily retrieved and then redeployed for optimal placement. Fluoroscopic and echocardiographic technologies are utilised to identify the positioning of the catheter.
Clinical trials on Amplatzer Piccolo Occluder
The long-term safety and effectiveness of the device were studied in a clinical trial named Amplatzer Duct Occluder II Additional Sizes IDE Study (NCT03055858). It was a single-arm, open-label, multi-centre study, which enrolled 200 patients across eight centres in the US.
The primary endpoint of the study was the rate of effective closure of the ductus arteriosus at six months and the rate of major adverse events through three years after the device implant.
Under the trial, the device was successfully implanted in 191 patients, leading to a 95.5% implant success rate. The primary endpoints of the study were met, with an effective closure rate of 100% and no major complications at three years.
A total of 156 of the available 182 patients (86%) completed the study. The survival rate exceeded 95%, with nine deaths reported. None of the deaths were determined to be related to the device or the procedure.
Patent ductus arteriosus
PDA occurs due to the formation of an opening between two blood vessels, originating from the heart. These vessels enable the transportation of oxygen-rich blood from the mother to the foetus for normal development.
The opening is automatically closed shortly after birth in most cases. In some premature babies, it fails to close on its own and causes increased blood flow to the lungs, causing breathing difficulties.
PDA constitutes up to 10% of all congenital heart disease cases. Urgent treatment is required for the survival of babies with PDA.
Marketing commentary on Abbott
Abbott is a US-based healthcare company focused on providing products in diagnostics, medical devices, nutrition and branded generic pharmaceuticals sectors.
The company employs 122,000 people and markets its products in more than 160 countries.