ACL TightRope® is a surgical implant system developed by Arthrex, a medical device company based in the US, for the treatment of orthopaedic injuries and anterior cruciate ligament (ACL) injuries in paediatric and young adolescent patients.
Arthrex, together with the orthopaedic surgeons of the Hospital for Special Surgery (HSS), developed all-epiphyseal and transphyseal techniques and instrumentation for skeletally immature patients for ACL surgery.
ACL TightRope implant system assists orthopaedic surgeons in carrying out performance-guided minimally invasive surgical procedures in young adolescents and athletes facing ACL injuries, based on their unique anatomy. The system received 510(k) clearance from the US Food and Drug Administration (FDA) in January 2023 for paediatric indications.
Details of ACL TightRope implant system
Arthrex’s ACL TightRope fixation devices portfolio includes the ACL TightRope implant, ACL TightRope II implant, ACL TightRope Attachable Button System (ABS) and implant, ACL TightRope DB, ACL TightRope RT graft fixation implant, FiberTag® TightRope implant, and ACL Repair TightRope implant with FiberRing™ sutures.
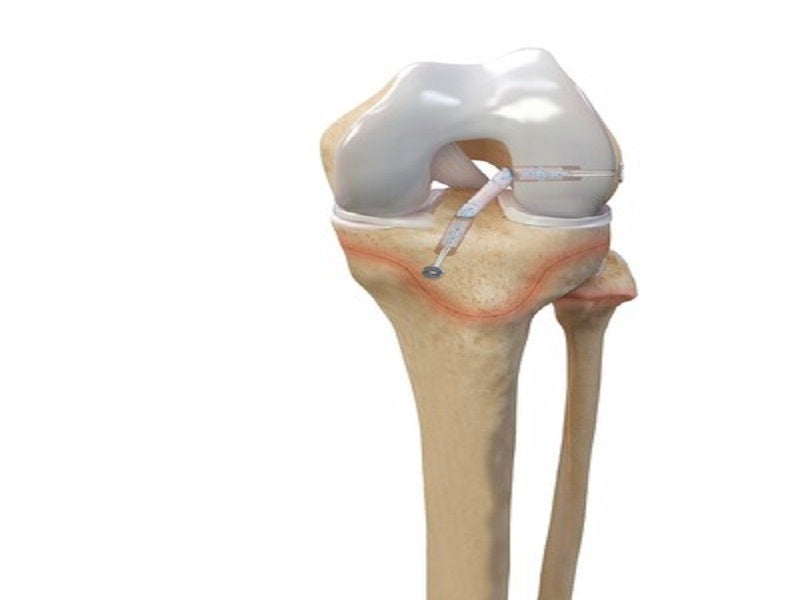
ACL TightRope comprises a suture loop that includes passing sutures and/or metallic buttons. The suture loop and passing sutures are braided nonabsorbable surgical sutures, while the button is made of titanium with holes to permit passage to suture and assembly with sutures.
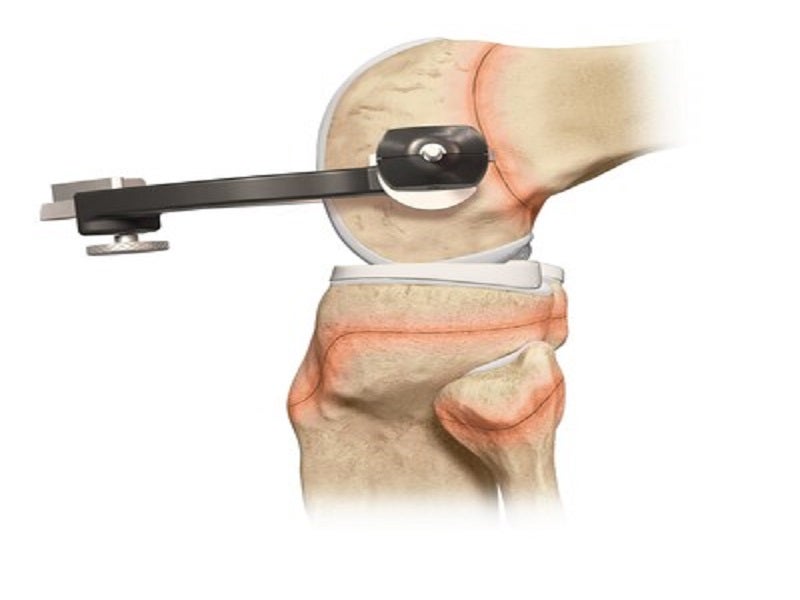
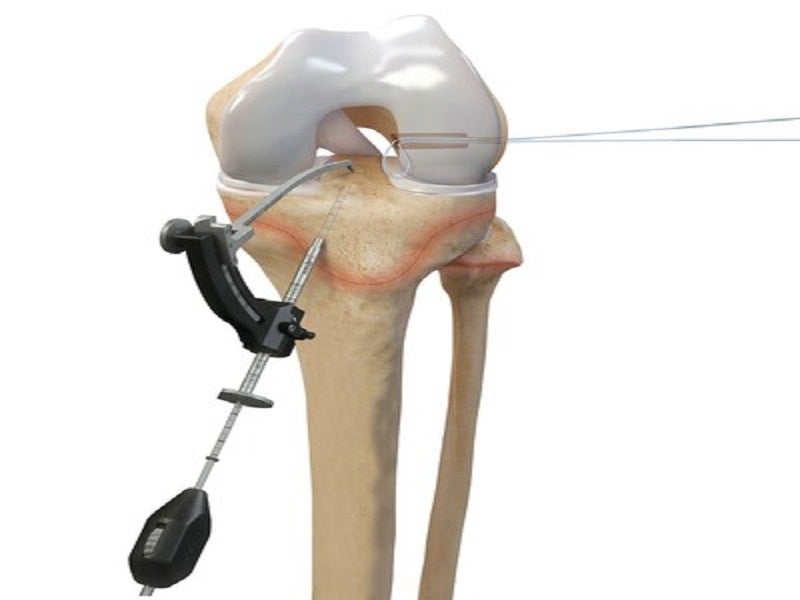
The paediatric-specific instrumentation and implants surgical technique guides enable surgeons to drill sockets and tunnels for the new, reconstructed ACL without affecting the growth plates, the areas of new bone growth in children.
The device helps surgeons drill across the growth plates to reconstruct the ACL using an all-soft-tissue autograft, in older adolescent patients who are approaching skeletal maturation. The paediatric and young-adolescent-specific guides significantly improve surgical options for reconstruction and epiphyseal fixation for ligamentous reconstruction or avulsion injury repair.
ACL TightRope implant is a four-point knotless fixation system, which provides adjustable cortical fixation for cruciate ligament reconstruction. The implant eliminates the need for multiple implant sizes and facilitates complete graft fill of short femoral sockets, which are common with anatomic ACL drilling.
ACL TightRope II features
The new ACL TightRope II is an adjustable-loop cortical suspensory fixation implant, which features a flat SutureTape design and a new proprietary fifth locking point used for fixation through the GraftLink® technique that offers the most in anatomic, minimally invasive, and reproducible ACL reconstruction.
ACL TightRope II implant offers superior biomechanics and better handling comfort to surgeons. It is more resistant to graft abrasion or tissue pull-through than traditional round sutures. The precise graft tensioning of the implant allows for incremental re-tensioning of the graft construct after final fixation.
The improved five-point locking button design resists cyclic displacement and easily accommodates the InternalBrace™ technique, which is associated with improved patient-reported outcome measures, less pain, and an earlier return to preinjury activity level.
The device is available in multiple configurations to accommodate various graft types and techniques, such as RT and BTB implants, loaded with an additional flipping suture or preloaded with FiberTape suture for the InternalBrace™ technique together with ABS and open ABS options.
ACL TightRope Attachable Button System (ABS) and implant
The TightRope ABS implant has improved the tibial fixation of ACL grafts used in knee ligament surgical procedures. TightRope ABS loops can be used on all graft types and attached to diverse configurations of the button for fixation over sockets created with a FlipCutter® III drill or full tunnels with concave buttons.
The new flat tape TightRope II ABS loop enhances handling and reduces graft abrasion. The two-piece TightRope II ABS implant swiftly passes through narrow bone tunnels and allows increased cortical fixation with the assembly of a large button over the cortical bone. The open TightRope II ABS implant was explicitly designed for closed-end grafts, such as BTB, Achilles tendon-bone blocks, or presutured QuadLink™, and GraftLink® allografts.
TightRope ABS implant benefits
TightRope ABS implant offers strong and reliable cortical fixation superior to interference screws and facilitates maximum graft-to-bone contact that improves incorporation and healing.
The implant provides the ability to retension grafts after fixation and knee cycling and comes with button options for sockets and full tunnels. The implant can close the tibial tunnel for preserving blood and growth factors in the tibial tunnel.
FiberTag® TightRope
The FiberTag® TightRope implant allows the attachment of single-ended grafts such as quad tendon grafts to the ACL TightRope RT and ABS implants. FiberTag® suture is integrated within the TightRope implant, enabling a strong and consistent connection between the suture and the TightRope loop.
ACL Repair TightRope implant
The ACL Repair TightRope implant is a knotless, tensionable system for ACL primary repair. It is an innovative approach to ACL preservation. It allows precise, incremental repair tensioning and re-tensioning. The new FiberRing™ suture enables simplified and reproducible suture passing and implant loading.
The ACL Repair TightRope implant comes preassembled with a FiberTape® suture to facilitate an InternalBrace™ technique.
FiberRing™ sutures come in 25mm, 35mm, 45mm, and 55mm lengths to adapt to various stitching techniques and the placement of multiple passes.