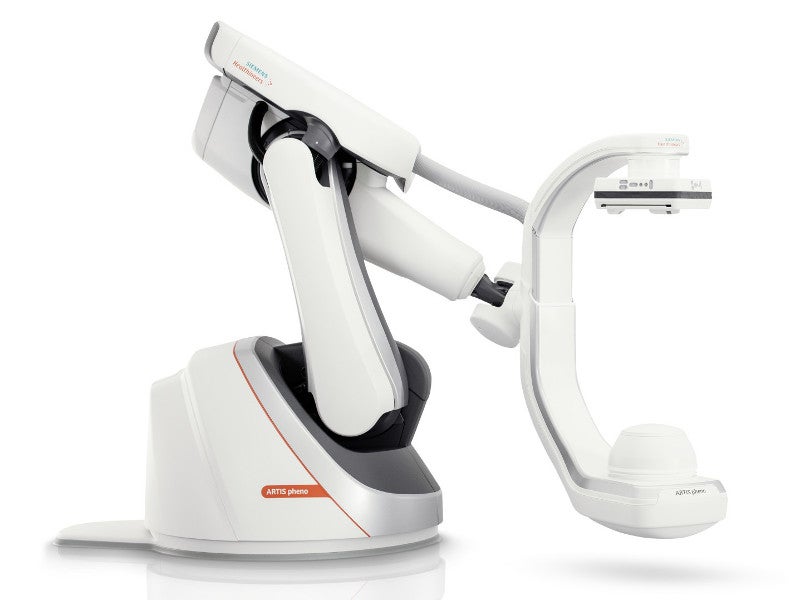
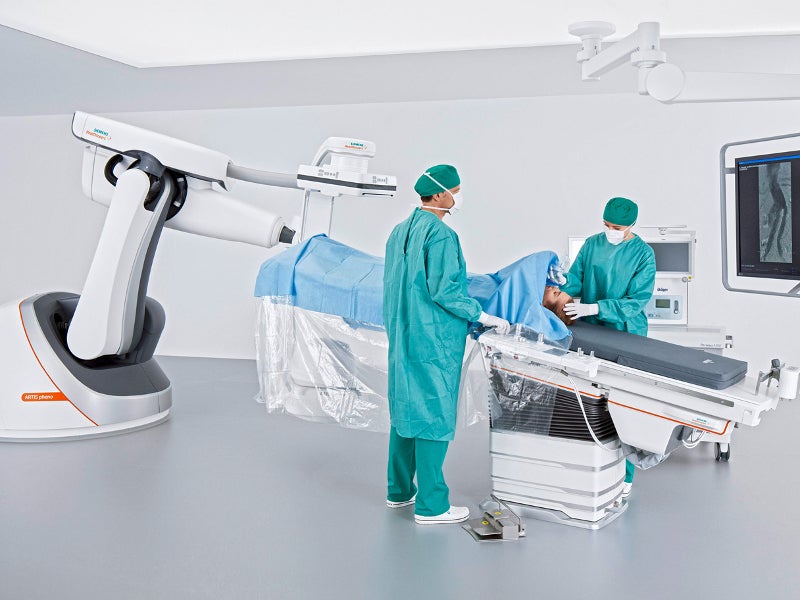
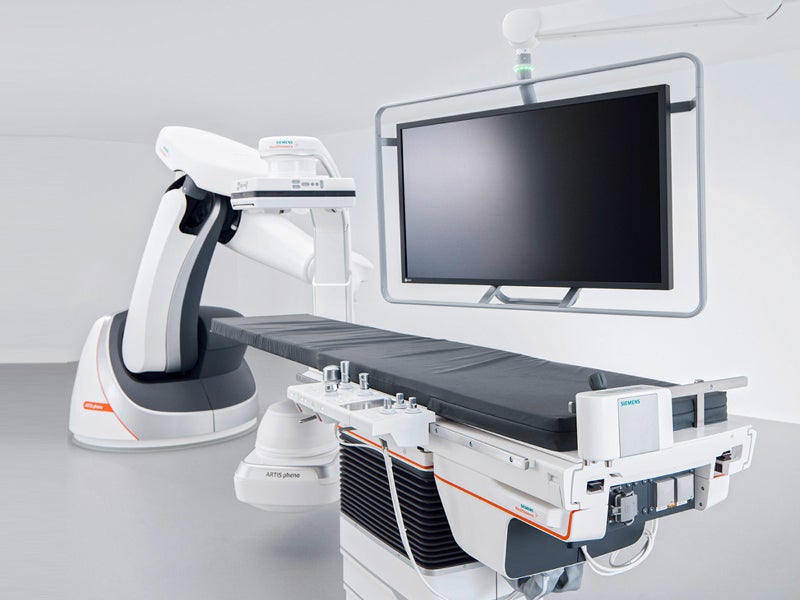
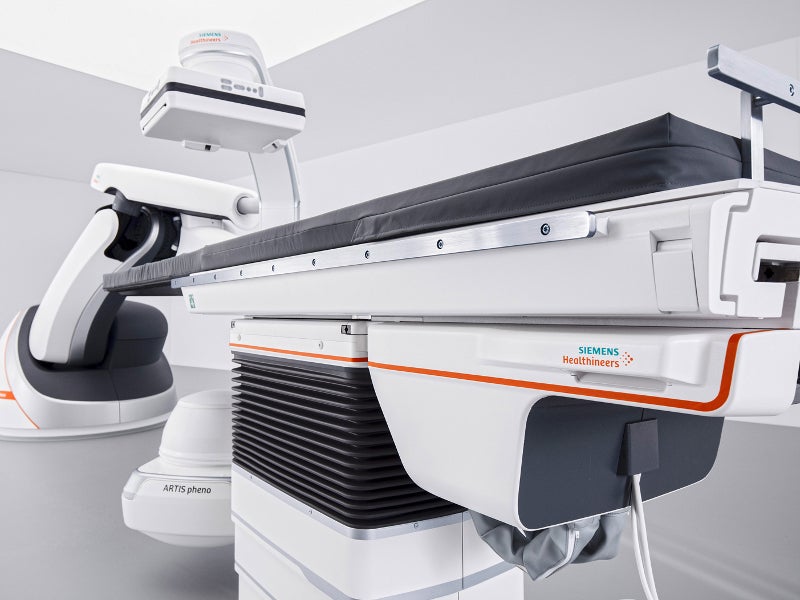
The ARTIS pheno angiography system is a robot-assisted device that is classified under the interventional fluoroscopic X-ray system by the US Food and Drug Administration (FDA).
The device was developed by Siemens Healthineers in collaboration with the at-design GbR studio.
Used for single and biplane diagnostic imaging and for interventional procedures, the device is equivalent to the previously marketed Siemens ARTIS Q and Q.zen modular angiographic system.
The ARTIS pheno angiography system received 510(k) certification from the FDA in March 2017. The device also won DesignEuropa Award in November 2018.
ARTIS pheno angiography system design and features
The ARTIS pheno angiography system is floor-mounted and comprises a C-arm, an X-ray tube, a digital imaging system, a digital detector and a collimator. The C-arm measures between 100cm and 130cm in diameter and has a free inner diameter of 95.5cm.
The system features an improved patient table, which can carry a patient load of up to 280kg. The motor-assisted table can be moved in longitudinal and transversal directions in levelled and tilted positions.
The tableside control modules of the system include the table control module (TCM), pilot control module (PCM) and collimator control module (CCM), which improve the movement of the table during procedure performance. The C-arm, stand and table surface are coated with anti-microbial paint.
The system also includes a Canon CSX-30 flat panel detector and one collimator for both angiography and cardiology.
The system has a 2k image matrix with up to 15 frames per second (f/s). The size of the display is 24in with an EIZO MX242W display.
The final image presentation is sharpened by automatic adjustment of contrast and brightness, improved noise reduction algorithm, and a spatial modulation transfer function of up to 16 frequency bands.
ARTIS pheno angiography system software details
The ARTIS pheno system is integrated with syngo application software VD20, which improves the functionality of the C-arm, table, tableside control modules, detector and collimator. It enhances the image processing and dose processing via Profinet protocol, which meets industrial standards.
The syngo DynaCT produces 3D images of organs with shorter scan times. DynaCT is an X-ray imaging software that converts 2D images obtained from a standard angiographic C-arm device into 3D.
The device also supports CO2 imaging of the extremities if the patient is sensitive to contrast agent and improves CO2 visibility using StructureScout feature. It includes Syngo Needle Guidance, which allows live and accurate visualisation of needle length and angle inside the body, ensuring that the procedure is performed precisely.
The system also features the ACUSON Freestyle Ultrasound System, which improves the viewing of ultrasound images. The CLEARstent Live stent enhancement software verifies the stent positioning in real-time during implantation.
ARTIS pheno angiography system benefits
The ARTIS pheno angiography system enables smooth performance of interventional procedures irrespective of size, condition and positioning requirements.
The easy-flow tabletop system enables positioning of the patient as it remains automatically aligned to the tabletop. The wider C-arm also improves the positioning flexibility of patients and provides enough room for the movement of complex imaging set-ups.
The device supports paravalvular leak closure and transarterial chemoembolization (TACE) of tumours. It is also claimed to be the first imaging system, which addresses the issue of hospital-acquired infections. The device facilitates efficient cleaning through internal cable guidance and helps combat infection.