The BrainScope One system is an electroencephalography (EEG) based medical device developed by BrainScope in partnership with the US Department of Defence.
The device records, measures, analyses and displays electrical activity in patients with traumatic brain injury.
The device was first approved for commercialisation under the name Ahead 300 by the US Food and Drug Administration (FDA) in September 2016. It was launched in January 2017 and received 510(k) certification from the FDA for clinical use in May 2018, when it was re-branded as BrainScope One.
In 2019, BrainScope received FDA clearance for additional labelling to include terminologies such as ‘multi-modal’ and ‘multi-parameter assessment of concussion and mild traumatic brain injury’.
BrainScope One EEG-based device design and features
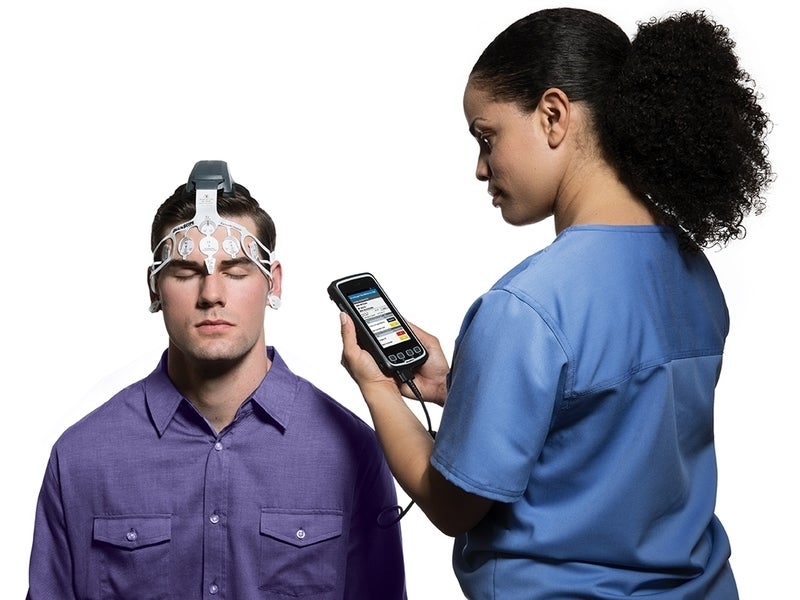
BrainScope One is a portable, non-invasive medical device that features BrainScope’s proprietary embedded software.
The system includes a disposable electrode headset that connects to a handheld display device, which uses state-of-the-art smartphone technology for recording data.
It uses algorithms and machine learning-powered EEG capabilities to analyse patient data and displays EEG in real time. The device records brain function parameters such as EEG and brain function index for the evaluation of human encephalogram.
The system also contains configurable, cognitive performance tests that provide a digital report to clinicians. It provides EEG playback and classifies structural injury.
In addition, the device displays raw and standard scores for cognitive performance. It records electronic versions of standard clinical assessments and generates an expansive list of library functions.
The headset comprises a silver/silver chloride (Ag/AgCl) electrode sensor array and a solid gel.
Technologies used in BrainScope One
BrainScope utilises the Trimble T41 mobile device. Direct current (DC) to 300Hz frequency bandwidth is used for processing 1kHz of sampled data. The system noise floor is less than 0.4 microvolts in 0.67Hz to 43Hz bandwidth.
The analogue-to-digital converter (ADC) resolution of the device is 45 nanovolt per bit with an ADC sampling rate of 1,000Hz. The ADC sampling rate could be downsampled to 100Hz for algorithm processing.
The device incorporates international 10-20 electrode placement system, which uses Fp1, Fp2, Fpz, AFz, F7, F8, A1, and A2 electrode positions.
BrainScope One EEG-based device benefits
BrainScope One has multi-modal capabilities to address a full spectrum of head injuries. It allows clinicians to take rapid, accurate and timely decisions for the patients at point-of-care. The device determines the prospect of concussion in patients along with its severity.
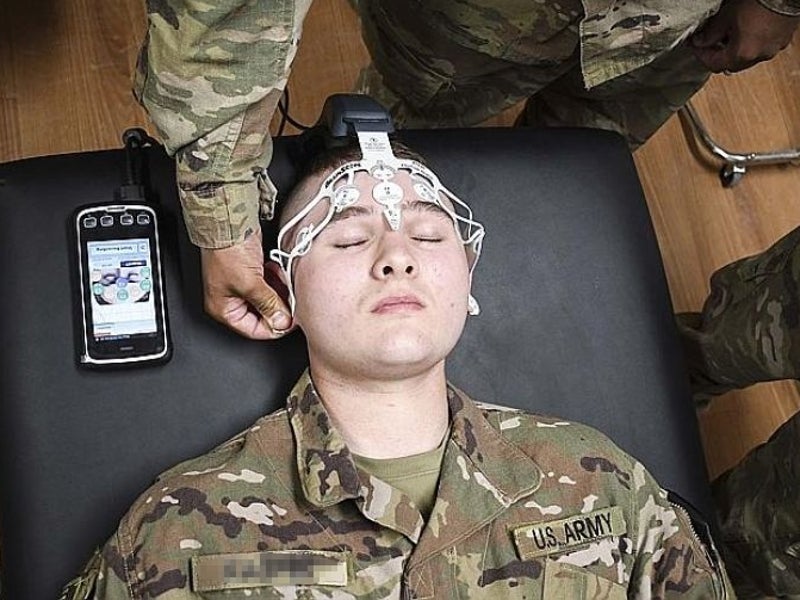
It facilitates US Army medics to assess military personnel, who have suffered a mild traumatic brain injury. The device can potentially cut down the usage of unrequired head CT scans to one-third in the emergency departments of hospitals thereby reducing healthcare costs by more than 30%.