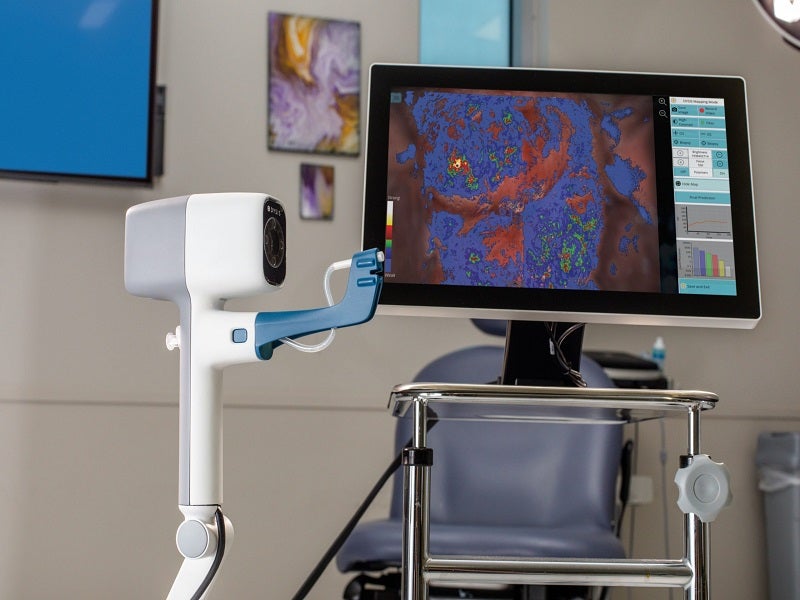
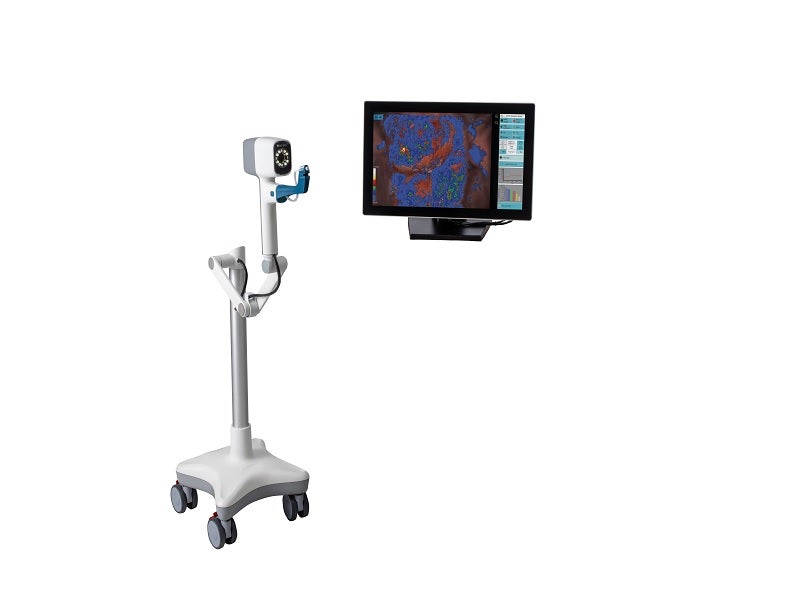
The DYSIS® View is a ready-to-use, compact, portable colposcope developed by medical technology company DYSIS Medical.
The device is designed to help healthcare professionals to detect cervical lesions more clearly and help direct their patient management decisions.
The DYSIS Colposcope was first cleared by the US Food and Drug Administration (FDA) for commercial use in March 2011 as a breakthrough device to assist clinicians in the non-invasive detection of cervical cancer as early as possible.
DYSIS with pseudo-colour imaging (PCI, or DYSISmap) as an adjunctive tool, is a digital colposcope designed to take images of the cervix under illumination and magnification.
The CE-marked device satisfies the European Medical Device Directive. It is also recommended by the UK National Institute for Health and Care Excellence (NICE) and recognised by the UK National Health Service’s (NHS) Cervical Screening Programme, Publication 20 guidelines.
Details and features of DYSIS View
The DYSIS View is an advanced colposcope with computer-aided cervical mapping. The system is equipped with a computer that runs the DYSIS proprietary software, containing DYSISmap™, a dynamic spectral imaging technology designed to objectively analyse aceto-whitening of the cervix.
The colposcope includes a 15-inch touchscreen monitor, a camera for high-resolution exam videos and images, green and high contrast image filters, live view magnification, digital biopsy markers, image capture, wi-fi connectivity, a patient database for record storage, and the ability to video and record biopsy or treatment procedures.
The device facilitates an instant replay during colposcopy exams, while the DYSIS SMARTtrack™ longitudinal tracking system allows a patient’s DYSIS View colposcopy exams to be compared with each other.
DYSISmap and DYSIS SMARTtrack
The new DYSISmap is a colour-coded map of the patient’s cervix’s aceto-whitening. It measures the onset, intensity and persistence of aceto-whitening and highlights the areas of the cervix with the most extreme whitening.
The information is intended to assist healthcare professionals in selecting the most appropriate areas for biopsy.
The map-assisted biopsies included in colposcopy examinations in the IMPROVE-COLPO trial increased the identification of patients with cervical intraepithelial neoplasia (CIN2+) or abnormal cervical cells by 44%.
DYSIS’ SMARTtrack software allows clinicians to track cervical lesions over time, which allows for a women’s cervical health to be conservatively managed.
DYSISviewer and SMARTtrack offer multidisciplinary collaboration with dynamic playback for post-examination, and multidisciplinary team (MDT) review to optimise patient treatment and effectively track cervical changes over time.
Benefits of DYSIS View
Colposcopy examinations conducted with DYSIS View are an effective approach to detect cervical abnormalities. The device has been shown to help healthcare professionals discover more high-grade disease on the first visit, increasing disease detection by more than 30% in all referrals and up to 50% in some patient groups.
Clinicians using the DYSIS colposcope can direct and manages patient appropriately and increases their confidence at the time of discharge while avoiding repeat examinations and treatment delays and lowering the risk of over-treatment. The device can fit right into any type of workflow.
Details of colposcopy
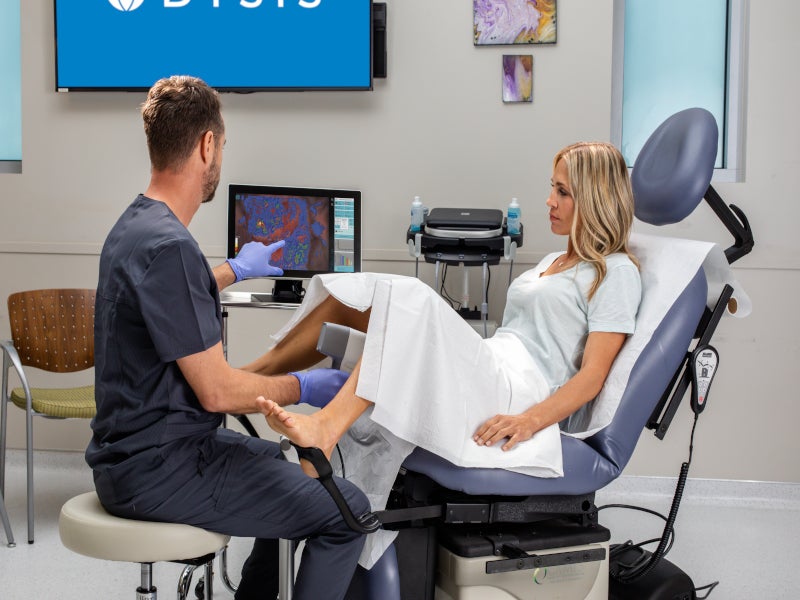
Colposcopy is a technique to examine the cervix and vagina using a colposcope, a special microscope with a light.
It is a non-invasive method in which a speculum is placed to keep the vaginal walls apart so the clinician can see the cervix correctly, similar to when a cervical screening test is taken.
A colposcopist applies a liquid solution such as acetic acid to the cervix to visually detect any regions of abnormal cells, which appear white. The examiner also looks for abnormal blood vessels and other associated abnormalities.
A colposcopy is typically not painful, although it may be uncomfortable. DYSIS colposcopy is an innovative approach to help healthcare professionals detect precancerous cervical lesions.