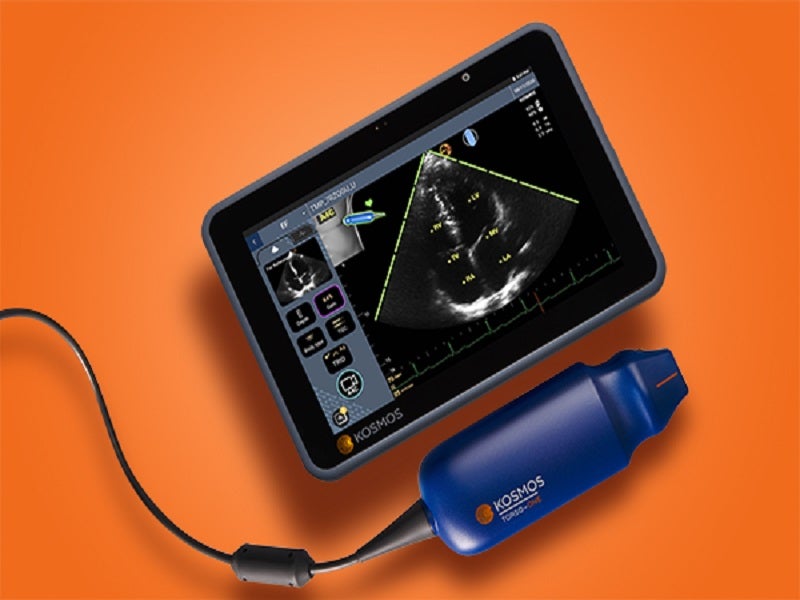
Kosmos® is a portable, AI-guided ultrasound scanner for high-resolution heart, lung, and abdomen imaging.
Developed by US-based medical equipment company EchoNous, Kosmos is the first patented hybrid point-of-care ultrasound (POCUS) clinical tool designed to boost diagnostic confidence at the bedside.
The scanner’s powerful imaging capabilities can identify a range of lung and cardiac disorders such as bilateral lung infiltrates, subpleural consolidations, irregular and thicker pleural lines, B line-like abnormalities and cardiac signs associated with myocardial dysfunction.
In September 2023, EchoNous expanded the system’s sophisticated features to be compatible with Apple iOS tablets, broadening the platform’s accessibility across diverse operating systems.
EchoNous unveiled Kosmos Plus, an enhanced version of the portable ultrasound device, in November 2023. Subsequently, in August 2024, the company released the Kosmos 2.2 update for Apple iOS, incorporating two new AI-powered functionalities, Auto Doppler for pulsed wave (PW) and tissue Doppler imaging (TDI), alongside Auto Preset for Torso One Transducer. These additions are designed to streamline the POCUS imaging process, making it more efficient and user-friendly for healthcare professionals.
Regulatory approvals for the Kosmos ultrasound system
The Kosmos platform became the first AI-assisted handheld ultrasound tool to receive US Food and Drug Administration (FDA) approval for clinical use in March 2020. It also received the European CE mark approval in the same month.
In August 2021, EchoNous’ Lexsa, a 128-channel linear probe designed for the Kosmos device, received FDA clearance for use in multiple healthcare specialities in the US. The probe allows Kosmos to deliver high-quality ultrasound images of vascular structures, nerves, and the musculoskeletal system.
The platform has been thoroughly tested in acute care clinical settings on more than 300 patients and in live clinical demonstrations by hundreds of physicians and imaging specialists from the US, Canada, Europe, and Japan.
Collaboration details
EchoNous collaborated with eInfochips, a product engineering and semiconductor design company based in the US, for the development of the cloud-based connected health Kosmos platform in May 2020.
The collaboration allowed the development of the world’s first AI-on-the-Edge ultrasound-based device together with deep learning for clinical assessment of the heart, lungs, and abdomen.
In August 2021, EchoNous partnered with Us2.ai, a software company based in Singapore, to offer accurate machine-learning-enabled heart health analysis for patients in minutes. EchoNous’ Kosmos platform integrated with Us2.ai’s software can provide automated analysis of heart structures that are scanned with Kosmos, enhancing accuracy and saving time.
EchoNous utilised Lantronix’s Open-Q™ Micro SOM (µSOM), development kit, and engineering services in the development of the Kosmos platform.
In August 2022, EchoNous partnered with Samsung, a South Korean manufacturing company, to enable medical professionals to run the Kosmos on off-the-shelf Galaxy Tab Active Pro tablets, supporting versions 12.0 and above.
EchoNous’ partnership with Israeli digital health pioneer UltraSight in June 2023 allows more medical professionals to conduct cardiac ultrasound in multiple care settings, improving patient access to cardiac monitoring. The UltraSight real-time AI guidance software, an AI-powered cardiac ultrasound technology, received FDA clearance in July 2023.
EchoNous entered into a collaborative agreement with United Medical Instruments, a medical device company, in alignment with the company’s strategic objectives to address increasing demand and bolster its market presence, in November 2023.
Kosmos portable ultrasound system design and specifications
Kosmos is mainly composed of the Kosmos Bridge display unit, a proprietary ultra-secure platform that runs the EchoNous system software, and the Kosmos Torso, an ultrasound imaging tool connected through a cable.
The Kosmos Bridge weighs 652g (23oz) while the Torso weighs 227g (8oz). The Torso device’s frequency range is between 50Hz and 60Hz while the display offers a high resolution of 1,920px × 1,200px.
The Bridge is a custom-designed Qualcomm® SDA835 system-on-chip (SoC) tablet that can conduct all AI computation functions by itself. It does not require cloud connectivity, ensuring patient and hospital data protection and no service interruptions during scanning.
The data can easily be shared with healthcare settings by seamlessly connecting to their information technology (IT) systems without internet connectivity. The tablet offers 128GB of internal storage and can also support remote storage.
The platform comes with three high-performance transducer probes with a high-durability piezoelectric-based phased array, including Torso, Torso-One and Lexsa. The Torso probe integrates the signals of a stethoscope, echocardiogram (ECG) and ultrasound in real-time. The handheld device offers continuous wave (CW) and PW Doppler capabilities.
The close-grip Torso-One probe occupies less space and offers high-definition ultrasound imaging while Lexsa provides superior performance with a 64/128 channel system in extremities, vascular and superficial applications. Both the Bridge and Torso devices are powered by batteries that can support 90 minutes of continuous scanning.
Kosmos ultrasound system features
The Kosmos device is the only portable ultrasound scanner that generates real-time ultrasound images using pulse-echo ultrasound. It ensures deep learning using high-grade image quality and algorithms for accurate results.
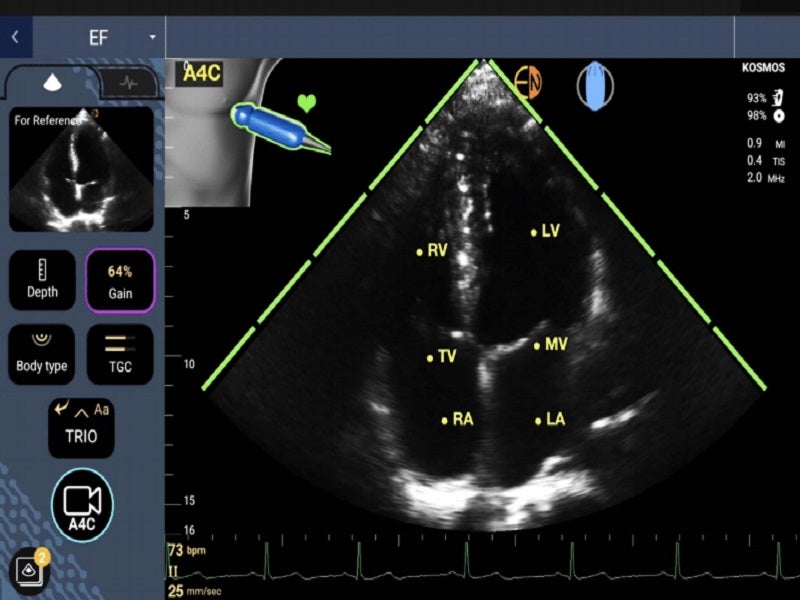
The platform also features a comprehensive suite of clinical deep learning applications planned for its 12-to-24-month roadmap. It displays its AI calculations to clinicians transparently, while performing clinical duties such as AI-calculated ejection fraction.
The portable probe and customised tablet can be moved quickly and easily around crowded hospital wards and the system’s long-lasting design makes it easy to disinfect.
The Kosmos app utilises Microsoft Azure’s cloud and its Internet of Things (IoT) capabilities to offer edge-to-cloud healthcare solutions.
Kosmos Plus design and features
The Kosmos Plus presents healthcare facilities with an economical substitute for high-priced ultrasound trolleys, without compromising the sophistication of features or the calibre of imaging.
Its user-friendly design ensures a seamless POCUS experience. The device is equipped with AI, encompassing advanced clinical functionalities and delivering diagnostic-grade imagery similar to top-tier cart-based systems.
It features state-of-the-art AI technologies such as Trio, AI FAST, and Auto EF, and superior Doppler capabilities, including PW, CW, and TDI, among others. It offers a variety of transducer choices, displays images on a 13in Apple iPad®, and comes complete with a medical-grade stand.
Kosmos 2.2 update on Apple iOS features
Kosmos 2.2 system’s Auto Doppler for PW and TDI is a first-of-its-kind feature. Utilising the system’s integrated AI technology, Auto Doppler automatically positions the Doppler sample gate at the ideal location for examining the selected valve. This advancement aims to streamline valve assessments by reducing manual system adjustments and lessening variability in gate placement. Consequently, clinicians can dedicate more attention to diagnostic insights rather than equipment settings.
Another intelligent functionality within the system is the Auto Preset. This feature dynamically adapts the examination preset according to the anatomy being scanned, transitioning smoothly between different body parts such as the abdomen, lungs, or heart. The system instinctively selects the most suitable preset for each scan, negating the need for manual adjustments and ensuring consistently high-quality imaging.
Additionally, the company has enhanced the workflow for the Auto EF feature by introducing a more sensitive Auto VTI trace capability, enabling more accuracy.
Benefits of the portable ultrasound system
Kosmos offers diagnostic-quality imaging for immediate clinical assessment at the patient’s bedside. It integrates ultrasound imaging with a three-lead, single-channel ECG and digital auscultation, enabling simultaneous evaluation of different aspects of a physiological event.
The device’s AI-assisted interface streamlines and enhances the image capture process. Its portability and ease of use make it ideal for various healthcare environments.
Kosmos not only delivers mobility and high processing capabilities but also comes with advantages such as early availability, technical support, and access to cost-effective development tools and resources.