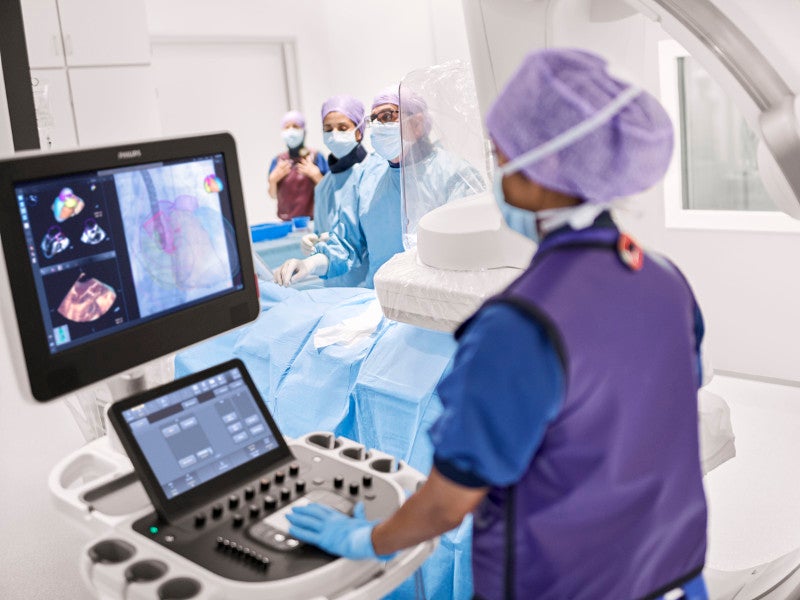
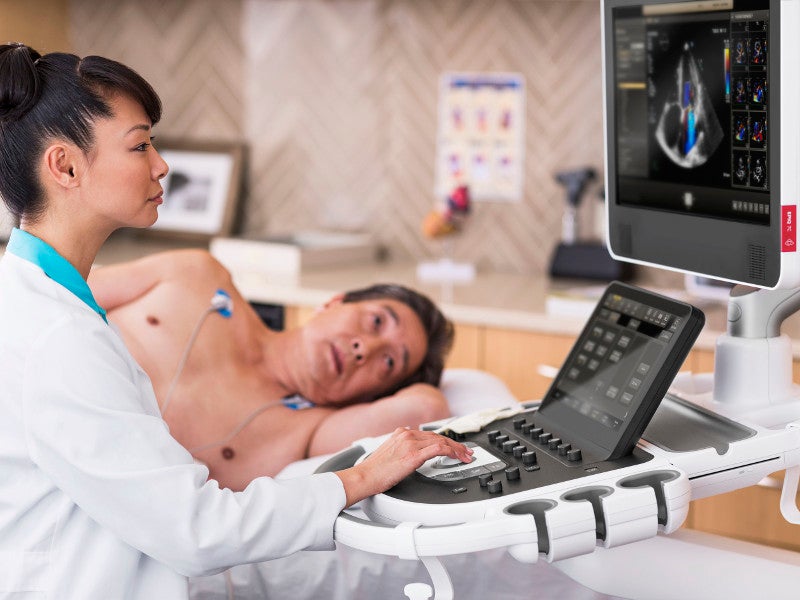
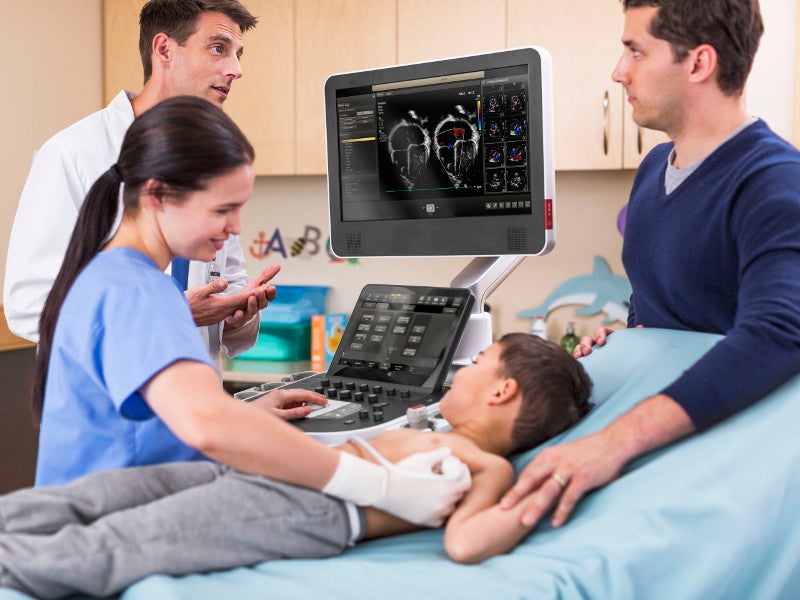
The EPIQ CVx is a software-controlled diagnostic cardiovascular ultrasound system developed by Philips based on the company’s Anatomical Intelligence Ultrasound (AIUS) platform.
The device is designed for diagnostic ultrasound imaging and fluid flow analysis for a wide range of indications, including cardiac, abdominal, musculoskeletal, urology, transvaginal and other small organs in foetal, paediatric and adult patients.
The ultrasound system received 510(k) certification from the US Food and Drug Administration (FDA) for clinical use in August 2018. The regulatory agency classified the system as an imaging pulsed Doppler ultrasonic system.
EPIQ CVx has also received CE mark in Europe.
EPIQ CVx cardiovascular ultrasound system design details
EPIQ CVx is a lightweight, portable ultrasound system weighing 104.3kg. It is 60.6cm wide, 109.2cm deep and 171.1cm high.
The system comprises a console and transducers. The console consists of a user interface, a 54.6cm display monitor with 180° movement, electronics and other peripheral entities.
The transducers are removable and connect to the system through multi-pin connectors using standard technology. The transducer frequency ranges from 1MHz-18MHz.
The system acoustic output limits include a spatial peak temporal average intensity (ISPTA max) of less than or equal to 720MW/cm², a mechanical index (MI max) of less than or equal to 1.9 and a thermal index less than or equal to 6.0.
The system contains 128 transmit channels and 256 receive channels. The various imaging modes of the system are B mode, M mode, PWD, CWD, Color Doppler, and combined modes.
Innovative features of EPIQ CVx
The EPIQ CVx ultrasound system features a configurable touchscreen user interface, which allows customisation and includes rotaries in a single row with fly-out controls. It includes the new TrueVue 3D photorealistic rendering, which enhances the viewing experience of the anatomical structures and improves the 3D workflow.
The device’s MultiVue real-time alignment solution contains live 3D cropping and MPR alignment tools, which minimise the risk of selecting a wrong device during interventional procedures. The system also features other 3D workflow tools such as QuickVue, FaceCrop, and AutoVue, which improve the efficiency of the system.
EPIQ CVx is equipped with Dynamic HeartModelA.I., which tracks each frame of a cardiac cycle using three-dimensional speckle technology, thus, improving the cardiac quantification.
Other tools of the ultrasound system
EPIQ CVx features EchoNavigator iXR integration, which allows real-time integration of EchoNavigator with fluoroscopy and Live 3D transesophageal echocardiography (TEE).
The PureWave paediatric S9-2 transducer has a frequency range of 2MHz-9MHz and a 15.36mm aperture. It offers 120° field view and is used for paediatric and adult cardiac applications. The curved array transducers C5-1 and C9-2 are useful for advanced foetal echo imaging.
The AutoSTRAIN feature allows for automatic detection and quantification of cardiac functions. The SmartExam reduces the examination time by 30%-50% and the number of keystrokes up to 300 per exam.
The ultrasound system also features Mitral Valve NavigatorA.I. for automated mitral valve measurement, Automated Cardiac Motion QuantificationA.I. for automated speckle analysis, and Automated 2D Cardiac QuantificationA.I..
Benefits of EPIQ CVx ultrasound system
The EPIQ CVx cardiovascular ultrasound system features second-generation OLED monitor with advanced visualisation techniques and quantification tools for crystal clear visual experience. The MaxVue provides ultrasound imaging in 16:9 high-definition and enlarges the anatomical images by 38% with no resolution loss compared to traditional ultrasound images.
The faster data processing, improved image quality, automated quantification, and tailored exam tools of the device simplify the clinicians’ workflow and reduce the requirement for repeat scans.
The system functions silently during a procedure with noise levels ranging between 37 decibels (db) and 41db, which is equivalent to the sound in a library. Further, it consumes 25% lesser power than traditional ultrasound systems.