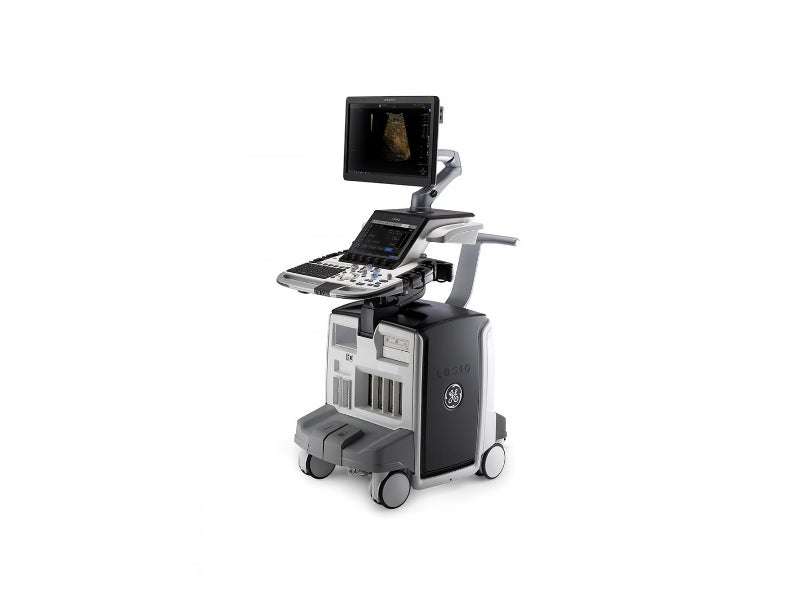
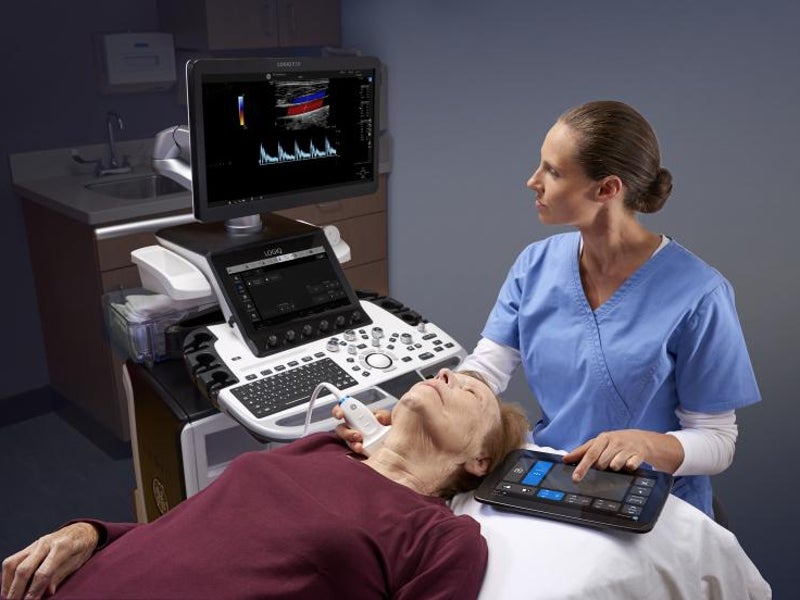
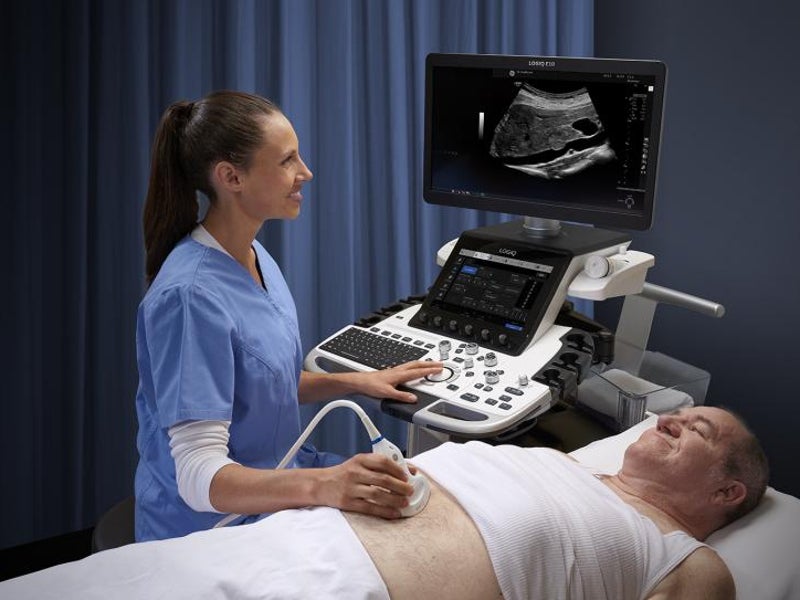
The LOGIQ E10 ultrasound system is an artificial intelligence (AI)-based radiology ultrasound system developed by GE Healthcare.
The device is designed to produce high-quality images for ultrasound evaluation of various body parts, including the liver, breast, abdomen, thyroid, testes and heart, as well as for diagnosis and treatment of patients for a wide range of conditions.
The ultrasound system received 510(k) certification from the US Food and Drug Administration (FDA) for clinical use in December 2017. The FDA classified the system as an imaging, pulsed Doppler and ultrasonic system.
LOGIQ E10 was commercially launched in February 2018.
LOGIQ E10 ultrasound system design and features
The LOGIQ E10 ultrasound system is a portable system comprising a mobile console with a width of 585mm, depth of 900mm and height of 1,300mm.
The ultrasound system features a 12in liquid crystal display (LCD) touchscreen with a colour wide-screen monitor, an adjustable floating computer keyboard, and specialised easy-to-reach controls.
The transducers and other materials that come in contact with the patient’s body are made up of biocompatible materials.
Advanced technologies used in LOGIQ E10 ultrasound system
The AI-powered LOGIQ E10 ultrasound system is a digital system that comprises the cSound™ Architecture technology platform to deliver high-quality images for diagnostic purposes.
It is equipped with an advanced graphics processing unit (GPU) and hardware technology that features 48 times more data throughput and ten-fold processing power than the substantially equivalent predicate device LOGIQ E9.
The device has the capability to reconstruct the acquired exam data in real-time. The XDclear™ probes of the device have broad bandwidth and function along with a cSound Imageformer for generating high-resolution images of even deep targets.
The system also features the Cloud-based communication platform Tricefy™, which allows for the integration of medical image sharing, remote viewing, and storage of an electronic health record (EHR).
The SonoDefense feature of the ultrasound system improves data security and offers a customisable solution for avoiding security breaches.
Imaging features of LOGIQ E10 ultrasound system
The ultrasound system features 2D shear wave elastography, which enables tissue elasticity to be evaluated quantitatively. The contrast-enhanced ultrasound enables sharp visualisation of tissue structure and lesion characterisation.
The system also includes 3D GPS or fusion imaging, which guides first needle positioning to reduce placement errors, examination time and stress. It is integrated with B-Steer+ and CrossXBeam features to improve the visual definition.
The B-Flow™ imaging feature of the device allows real-time viewing of blood flow echoes, while the power Doppler imaging (PDI) sensitivity allows detection of slow movement of blood even in small blood vessels.
The LOGIQview feature of the device allows panoramic viewing of images up to 60cm. The split screen feature enables side-by-side viewing of images with or without colour and power Doppler for efficient comparisons.
The digital technologies of the system improve the processing power, enhancing data throughput for delivering clearer images.
Other features of LOGIQ E10 ultrasound system
The Photo Assistant application allows clinicians to photograph swelling or ulcerations during a study. The photographs provide valuable insights for diagnostic and treatment purposes.
The remote control application enables remote operation of the system, while improving the workflow, efficiency, and patient experience.
The Scan Assistant feature allows customisation of the automation according to the user for improving their efficiency, while Compare Assistant enables comparison of the patient’s previous study with the latest ones.