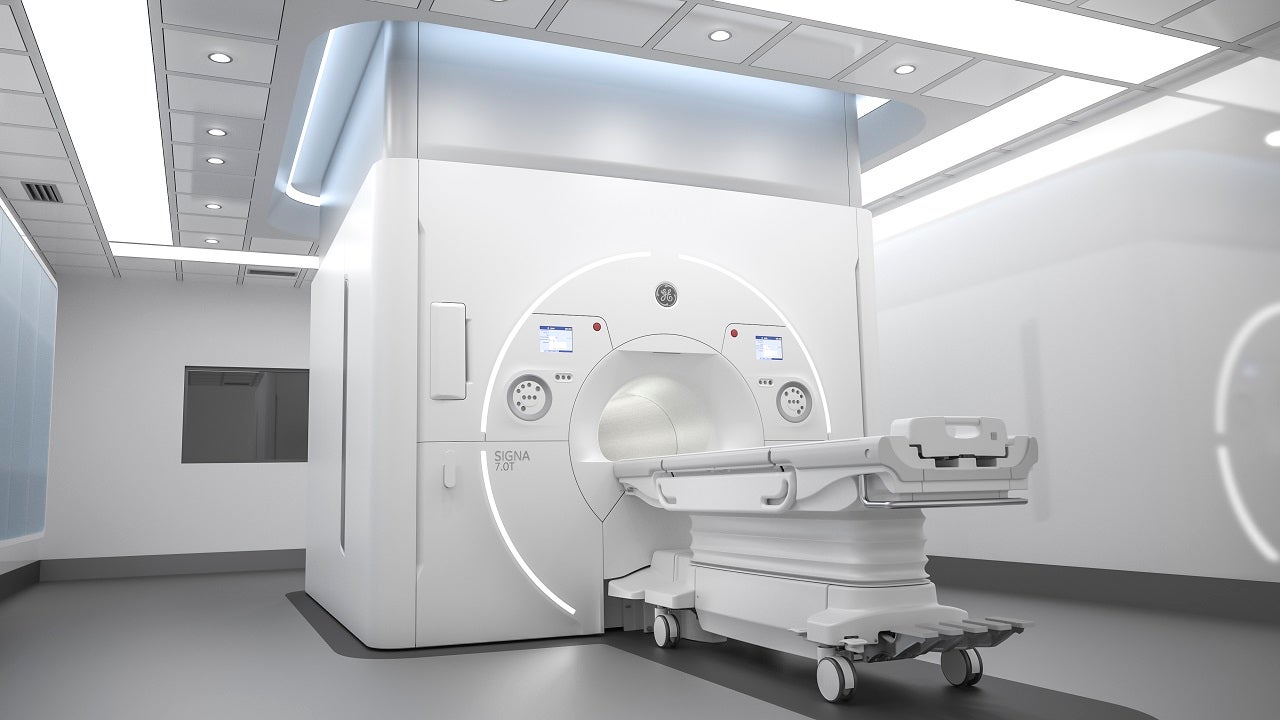
SIGNA 7.0T is a magnetic resonance imaging (MRI) scanner developed by GE Healthcare for neurological and musculoskeletal imaging functions.
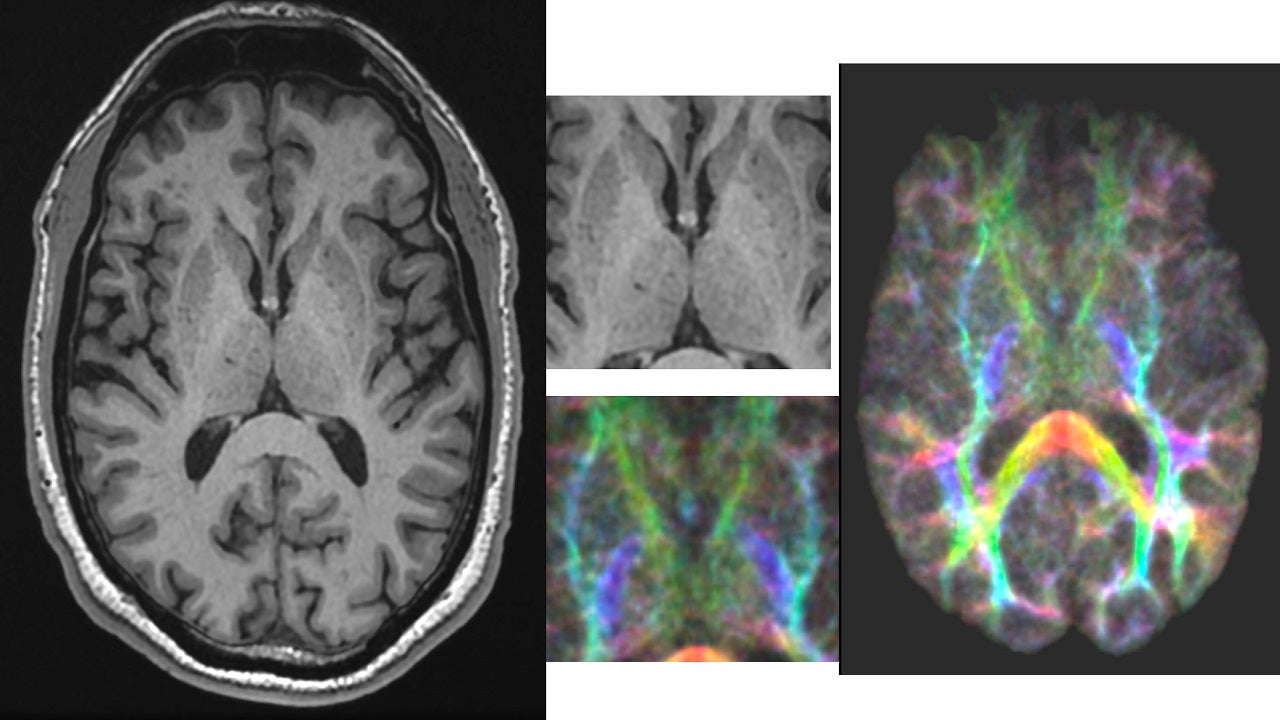
The device will be utilised for high-resolution, detailed anatomical, functional, metabolism and microvasculature imaging of the brain and joints for research and clinical purposes. It will also be a crucial tool for research into psychiatric disorders such as Alzheimer’s disease and mild traumatic brain injury.
The US Food and Drug Administration (FDA) cleared the device for human use in November 2020.
GE’s SIGNA 7.0T MRI scanner features
The SIGNA 7.0T MRI scanner features a 60cm-wide bore with UltraG gradient magnet technology within its core.
UltraG is a full-body gradient coil designed to meet the requirements of ultra-high-field imaging. It has a hollow conductor and a water-cooled design with more than 45kW heat dissipation. The gradient coil is characterised with 113mT / m peak amplitude per axis and 260T / ms peak slew rate per axis.
The SIGNA 7.0T MRI scanner incorporates GE’s new SIGNA™Works software framework with state-of-the-art applications including deep learning capabilities such as AIRx™ for automatic slice positioning and silent MR imaging, which allows smooth protocol translation between GE MR Systems.
AIRx is an automatic slice prescription used for brain scanning, that allows five times faster set-up time with four times fewer mouse clicks.
Additionally, the scanner is integrated with a Precision RF transmit. The architecture enables development in parallel transmit, improved image quality, and flexibility in research.
The device also offers a revolutionary platform for advanced knee and cartilage imaging, facilitating anatomical simulation in ultra-high-resolution as well as research capabilities to assess quantitative changes related to disease in anatomy. It will support neurodegenerative disease and extremities imaging as well.
SIGNA™Works platform details
SIGNA™Works is a completely customisable platform that is integrated with SIGNA™ Architect solution. It improves the precision, consistency and quality of imaging while enhancing the patient experience with silent, contrast-free, free-breathing and needle-free applications.
The technology can be upgraded and customised to satisfy the requirements of the growing practice with additional applications.
The standard and elective applications of SIGNA™Works are BodyWorks, CVWorks, NeuroWorks, OncoWorks, OrthoWorks, and PaedWorks. The innovative applications include HyperWorks, ImageWorks, SilentWorks and ViosWorks.
SilentWorks is the most advanced noise-reducing technology of GE. It is available across all anatomies and can be utilised for multiple weights and coils. ViosWorks, powered by Arterys™, improves cardiac MR assessment by capturing all seven dimensions of information related to the cardiovascular scan in approximately ten minutes.
The software can extend the clinical experience with better image quality, improved performance, and a more efficient workflow. SIGNA™Works optimises operation and throughput with decreased scanning times.
Marketing commentary on GE Healthcare
GE Healthcare is a medical device company based in the US. The company’s intelligent devices, applications and services supported by Edison intelligence platform allow physicians to make quicker and more informed choices.
The company offers medical technology, digital infrastructure, data analytics, and decision support resources to help healthcare practitioners diagnose, treat, and track their patients. It also offers services, accessories, consumables, education, training and consulting.
The company employs approximately 50,000 people and operates in more than 160 countries. It has installed approximately four million imaging, mobile diagnostic and monitoring units globally that generate more than two billion scans a year.