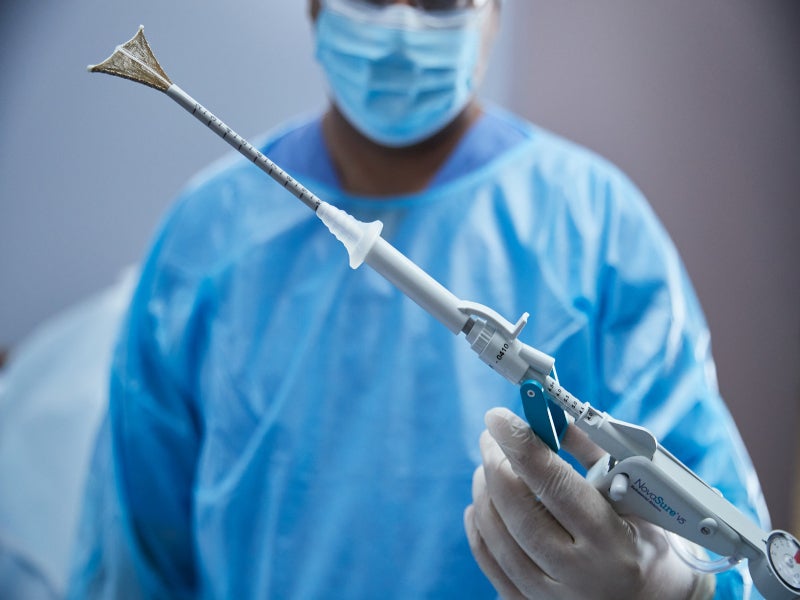
NovaSure V5 is an innovative global endometrial ablation (GEA) device developed by the US-based medical technology company Hologic to help surgeons in endometrial ablation procedures for the treatment of abnormal uterine bleeding in women.
NovaSure V5 is a newer version of the existing 8mm-diameter NovaSure device, incorporating improved features to treat a wide range of cervical and uterine anatomies.
NovaSure V5 was launched in the US in November 2021. It received approval for use in Canada and Europe in February 2023 while its previous version was approved by the US Food and Drug Administration and Health Canada in 2001.
The device delivers radiofrequency (RF) energy to ablate the endometrial lining of the uterus to reduce or stop abnormal menstrual bleeding in women. The device has benefitted more than three million patients since its approval.
NovaSure V5 GEA device design – details
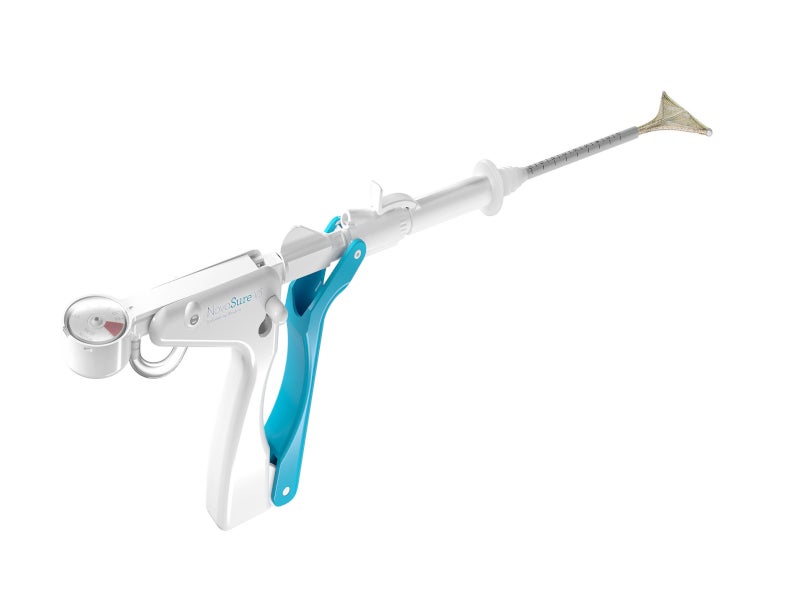
The next-generation NovaSure V5 GEA device is an impedance-controlled endometrial ablation system with a more patient-centric design for a more customised ablation during each procedure. It consists of the NovaSure disposable device with a connecting cord, a NovaSure RF controller, a NovaSure CO₂ canister, a desiccant, a foot switch and a power cord.
The NovaSure disposable device contains four uniquely designed electrodes arranged in an array mounted on an expandable frame to deliver adequate bipolar RF energy up to the appropriate depth on the entire interior surface area of the uterine cavity. The device is inserted trans-cervically into the uterine cavity, where the slender 6mm diameter sheath with AccuSheath™ markings retracts to allow the bipolar electrode array to be deployed and conform to the uterine cavity.
The disposable device works in conjunction with a dedicated NovaSure RF controller, which automatically calculates the optimal power level required for the treatment of the uterine cavity, based on the uterine size. It maintains pressure at 50mmHg for a minimum of four seconds to keep the uterine cavity intact and stops the procedure once 50ohms of impedance has been reached or after two minutes.
The UNIQUE SureClear™ Technology fluid removal system offers constant tissue contact with the array through integrated suction while removing the steam, moisture and other ablation by-products from the uterus simultaneously.
The NovaSure CO₂ canister, desiccant, and foot switch are connected to the RF controller for different functions. The foot switch is used to activate the NovaSure RF controller. The CO₂ is used by the built-in cavity integrity assessment system to pressurise the uterine cavity for the assessment of uterine cavity integrity. The desiccant absorbs the moisture removed from the uterine cavity through the suction tubing during the ablation procedure.
The average treatment time is 90 seconds and does not take beyond two minutes. The system can be used in hospital outpatient, inpatient, emergency and ambulatory settings with safety and efficacy.
The technologies involved
NovaSure V5 uses SmartDepth™ technology for endometrial ablation, which offers a more customised, reliable and reproducible depth of ablation to each patient.
The new V5 cervical seal of the system utilises EndoForm™ technology to help patients with a large cervix. It is designed to increase the device’s sealing surface and accommodates a range of cervical canals and anatomies.
The UNIQUE SureClear™ technology offers the feature of integrated suction and removal of ablation products to the device.
The device’s AccuSheath™ markings improve seating and fundal placement accuracy and confidence. The new V5 depth markings allow the 6mm sheath to confirm the fundal placement and provide more certainty on the exact placement of the device in the uterine cavity.
Advantages of the NovaSure V5 GEA device
The device has achieved 97% patient satisfaction and a 75% amenorrhea rate after five years. Approximately 86% of patients treated using NovaSure avoided hysterectomy after ten years.
The device is also effective in the treatment of painful and haemorrhagic symptoms associated with adenomyosis in both the short and long term.
The device is equipped not only to improve patients’ comfort and ease of use for the physician but is also designed to be suitable for various cervical canal sizes while maintaining the clinical efficacy of the NovaSure system. It eliminates the need to scan the myometrial thickness before treating patients with NovaSure.
The NovaSure RF technology tapers the depth of ablation of the uterine cavity for sufficient penetration into the myometrium for consistent results irrespective of the patient’s cavity size.