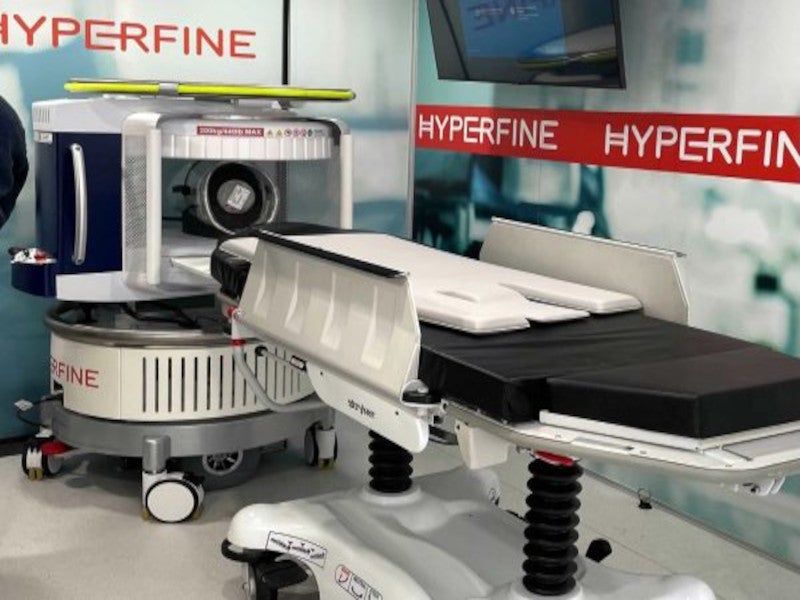
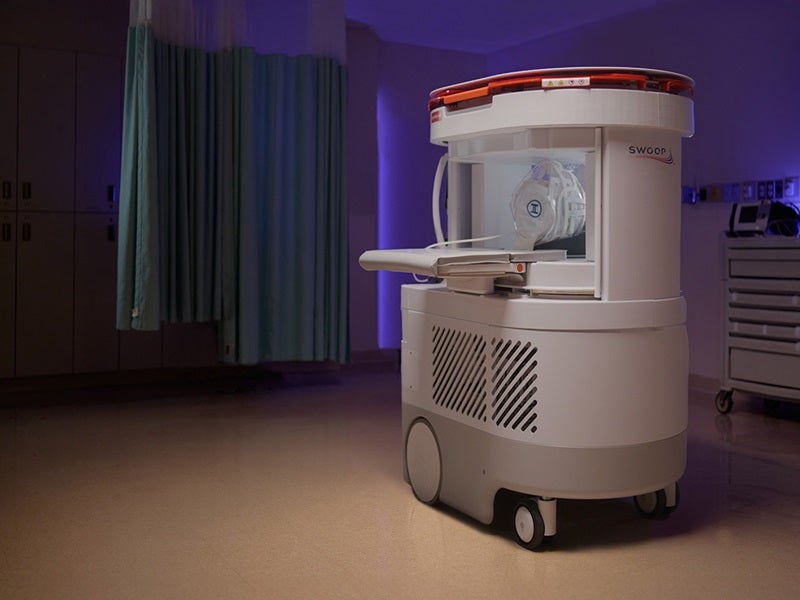
The Swoop® system is a first-of-its-kind portable, ultra-low-field magnetic resonance imaging (MRI) device for point-of-care settings such as intensive care units, emergency departments, mobile stroke units and resource-limited environments.
Developed by Hyperfine Research, a medical technology company based in the US, the system is suitable for general brain imaging, headaches, stroke, non-specific muscular weakness and encephalopathy. It can monitor coronavirus (Covid-19) patients, who are known to be at risk of neurological disorders.
In April 2021, the Aga Khan University’s Medical College in Karachi, Pakistan, became the first country in Asia to acquire the portable Swoop MRI system. The system was installed in the UC Davis Health in North California in the US for diagnosing traumatic brain injuries in March 2022, followed by its introduction in the Jefferson Abington Hospital in Pennsylvania in June 2023.
In July 2025, Hyperfine released the Optive AI™ software for existing Swoop® system customers in the US, following US Food and Drug Administration (FDA) clearance in May 2025.
Hyperfine gained FDA clearance for a new portable MRI scanner that uses the Optive AI™ software in June 2025.
In September 2025, Hyperfine secured CE marking and UK Conformity Assessed (UKCA) approval for the Optive AI™ software.
The Swoop® system is available in various markets including India, Indonesia and Singapore, Israel, Malaysia, New Zealand, Pakistan, Saudi Arabia and Türkiye.
Swoop portable MRI system approvals
Swoop received 510(k) clearance from the FDA for MRI of the brain and head of patients of all ages, including patients from birth to two years, in August 2020.
The approval came after user feedback and technology advancements had been incorporated into the original device, which was cleared in February 2020.
The FDA cleared Hyperfine’s deep learning image analysis software, Advanced AI Applications, in January 2021. The image analysis software measures brain structure and pathology in images acquired by the Swoop MRI system.
The additional clearance made the system a true bedside MRI platform, supporting efficient clinical decision-making for assessing brain health and injury. The FDA also granted clearance for the updated AI-powered software of the Swoop system to improve diffusion-weighted imaging (DWI) significantly.
The update expands Hyperfine’s AI denoising capabilities within the system, incorporating advanced image post-processing specifically into the DWI sequence. Previously utilised for sequences such as T1, T2 and fluid-attenuated inversion recovery (FLAIR), this AI feature now enhances the clarity of images, potentially improving clinicians’ ability to diagnose, treat and monitor patients undergoing brain imaging.
The Swoop MRI system holds Conformité Européenne (CE) certification in Europe. The system has been approved for brain imaging in many countries, including Canada and Australia. It has UKCA certification in the UK and is also available in New Zealand and Pakistan.
Hyperfine also achieved CE certification under the European Union Medical Device Regulation (EU MDR) and obtained UKCA certification in the UK for the latest hardware and eighth-generation AI-powered software in 2023.
Details and specifications of Hyperfine’s Swoop
Swoop is a lightweight, compact, easily movable portable MRI system measuring 140cm high and 86cm wide.
Weighing 630kg, the system can be transported on wheels and easily moved through standard doorways and elevators. It can be wheeled directly to the patient’s bedside, plugged into a standard electrical wall outlet and controlled through an easy-to-use wireless tablet such as a 12.9in Apple iPad®.
Its portability allows healthcare providers to bring MRI capabilities to emergency departments, intensive care units and rural or underserved locations where access to traditional MRI machines may be limited.
The magnet design and field strength of 0.064 Tesla reduces exposure risk for staff, patients and family members.
Designed to complement standard MRI systems, Swoop lets new users receive training in device navigation, operation and safety in just 30 minutes, allowing clinicians to streamline workflow.
Swoop portable MRI system features
The system features in-built noise cancellation technology that maps and adjusts the high electromagnetic interference in the environment, resulting in crisp, clear T1-weighted, T2-weighted, DWI and FLAIR sequences.
One of the key features of the Swoop system is its simplicity and ease of use.
The system is operated through an intuitive touchscreen interface, making it accessible to healthcare professionals with minimal training.
The scanning process can be initiated quickly, and the system guides users through the necessary steps to acquire high-quality images.
Its portability eliminates the need for patient transportation, reducing delays in diagnosis and treatment.
The Swoop system is designed to be energy-efficient, consuming significantly less power than traditional MRI machines.
The reduced power requirement not only lowers operating costs but also minimises the environmental impact associated with energy consumption.
Advanced AI applications details
Advanced AI applications form part of Swoop’s standard software package, supporting decision-making and ensuring rapid feedback for diagnostic insight.
The applications use tools for measurement, including automated ventricular volume measurement, brain extraction, brain alignment and midline displacement.
The Optive AI™ software is a tenth-generation update that modifies multiple stages of the imaging workflow for the Swoop system, including noise reduction, image capture, reconstruction and post-processing.
The software has been developed to provide improved image quality, functionality and ease of use for clinical brain imaging.
In March 2025, Hyperfine announced a partnership with Nvidia to enhance AI-driven image reconstruction and integrate real-time clinical decision support within the portable MRI workflow.
Utilising Nvidia’s comprehensive training and inference tools, including DALI and MONAI, the partnership aims to improve the Swoop® system’s image quality, shorten scan durations and support quicker, more dependable diagnoses.
Benefits of the Swoop portable MRI system
The Swoop portable MRI system reduces acquisition costs by 20 times, consumes 35 times less power and weighs a tenth of existing fixed conventional MRI systems.
It provides patients and clinicians with high accessibility, immediacy and convenience.
Swoop overcomes the constraints of existing imaging technologies and makes MRI available to every patient, at any time and in any place.
The patient-friendly open layout of the system reduces patient anxiety. It displays images in minutes, allowing for critical decision-making in clinical settings such as neuro-intensive care units, emergency rooms and paediatric settings. It also allows physicians to move more swiftly from scans to suggested courses of treatment.
Clinical research details using Swoop portable MRI system
The Swoop portable MRI system is being used in global clinical research studies. In May 2023, Hyperfine expanded its clinical research, funded by the Bill & Melinda Gates Foundation, to investigate the neurological effects of early childhood malnutrition in low and middle-income countries. The initiative aims to understand the impact of malnutrition on children’s school readiness and future academic success.
A $3m grant from the foundation extends this research over three years, during which Hyperfine plans to enhance the Swoop portable MRI system, to improve its functionality, software and data management for remote settings. The grant also supports the development and optimisation of neuroimaging MRI sequences for neonates and infants.
The system is used in the CARE PMR observational study to evaluate the clinical utility and workflow benefits of Swoop system images in detecting amyloid-related imaging abnormalities in Alzheimer’s patients receiving amyloid-targeting therapy.
Hyperfine collaborated with Athletic Heart Cardiac and Metabolic Testing, a mobile medical diagnostic company, to provide portable brain imaging screenings to current and retired professional athletes, starting with retired professional basketball players.
Investigators of the ACTION PMR [post-marketing requirement] study aim to integrate brain imaging with the Swoop system into the stroke diagnosis and treatment workflow in an international, multi-site prospective study.
The system is also used in the HOPE PMR observational study to enhance the standard of care for paediatric hydrocephalus management.