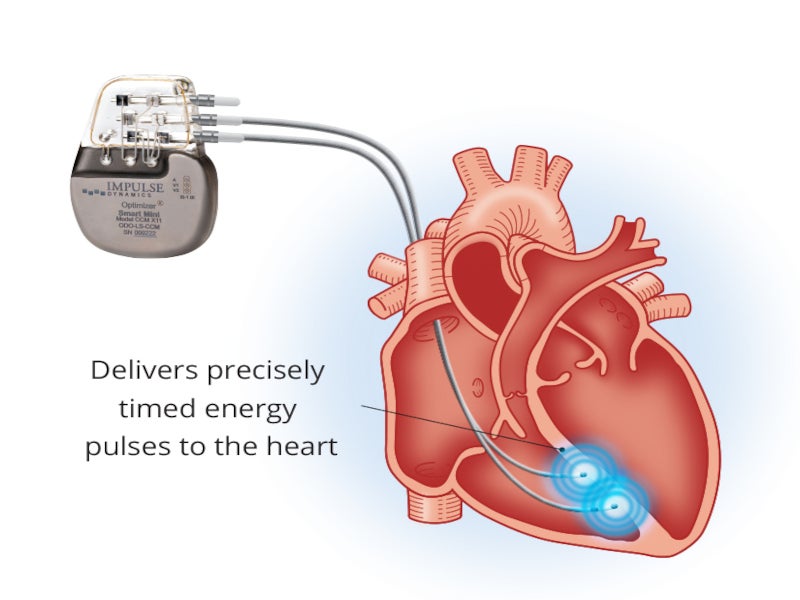
The Optimizer® Smart system is a first-of-its-kind, minimally invasive implantable cardiac contractility modulation (CCM) therapy delivery device developed by medical device company Impulse Dynamics.
It is the first-approved CCM device for the treatment of moderate to severe chronic heart failure in patients who remain at risk despite receiving medical treatment.
It received breakthrough device designation from the US Food and Drug Administration (FDA) in 2015. The FDA provided clearance for its clinical use in March 2019. The first patient was implanted with the device at the Ohio State University Wexner Medical Centre in May 2019.
In December 2020, Impulse Dynamics secured $60m in Series D crossover financing led by the CR-CCT Industry Pilot Fund, healthcare investment firm Redmile Group, and Perceptive Advisors, an investment management firm focused on the life sciences industry. The CR-CCT Industry Pilot Fund is co-managed by China Resources Capital Management and China Chengtong (HK). A portion of the proceeds from the funds was used for the commercialisation of the Optimizer® Smart implantable device in the US.
Approvals and launches
Impulse Dynamics received approval from the FDA to modify the Optimizer® Smart medical device’s labelling in October 2021. The approval allows the company to remove normal sinus rhythm, which previously served as a barrier to CCM therapy concerned with heart rhythm due to the indications for use statement. The labelling modification substantially increases the number of beneficiaries of CCM therapy.
In April 2022, the company launched the Optimizer® Smart’s successor Optimizer® Smart Mini CCM delivery system and subsequently completed its first implant in the US in May 2022.
The Optimizer® Smart Mini CCM® therapy delivery system received the FDA and CE Mark approval in April 2022. The first implant of the device in Europe was carried out in Italy in June 2022.
More than 7,000 patients have been treated using the CCM therapy in 44 countries and regions including Australia, Brazil, China, Europe, Hong Kong, India and New Zealand.
Optimizer smart system design and features
The Optimizer® smart system comprises an Optimizer® smart pulse generator, an OMNI II Programmer, and a mini charger.
The rechargeable Optimizer pulse generator is easily implanted into the patient’s heart with two lead configurations. The charge coil and telemetry functions are integrated into the system for flexible positioning of the device into the heart. The device is powered by a Quallion-manufactured Model QL0200I-A lithium-ion battery with 0.2Ah usable capacity.
OMNI II Programmer is a touchscreen tablet or laptop that features the Optimizer smart programmer application software, connected to a programmer interface. A magnetic induction telemetry wand is placed over the implant location to establish communication between the pulse generator and the programmer.
The graphical user interface enables rapid programming, allowing the software to perform a remote operation. The programmer is also equipped with a mini-Bluetooth printer, providing access to all crucial device data at once.
The mini charger can charge the device within 40 to 60 minutes, featuring a light-emitting diode display and a single button to avoid any human error. The charger does not require a power line connection during charging.
Optimizer Smart Mini System design and features
The next-generation Optimizer Smart Mini system comprises an implantable stimulating device the size of a pacemaker, and an external charging port.
The device has a rechargeable battery that can last up to 20 years. It enables diagnostic monitoring for heart failure, offering valuable clinical insights to healthcare providers, aiding them in the management of patients with the condition.
The device is 25% smaller and 33% lighter than its predecessor. It is easier to implant and more comfortable for patients.
Details of the cardiac contractility modulation therapy
CCM is a novel therapy in which the patient’s cardiac muscle is electrically stimulated during the absolute refractory period. Unlike other implantable systems such as pacemakers, CCM therapy modulates the heart muscle strength without affecting heartbeat or action potential distribution.
The electrical impulses trigger physiological processes in the heart’s muscular cells, which create an impact on cellular function at the molecular level and improve heart function. The therapy is intended to enhance the patient’s physical ability and well-being.
CCM therapy has been clinically evaluated in 2,000 patients.
Optimizer smart system implantation procedure
The Optimizer® device is implanted in the pectoral region on the right side of the patient at a maximum depth of 4cm and connected to two conventional pacemaker leads positioned on the ventricular septum at about 2cm. The leads are attached to the right ventricle through veins, with one tracking ventricular activity and the other tracking atrial activity.
The leads deliver electric CCM signals to the heart’s right ventricle at regular intervals throughout the day.
Clinical studies on the Optimizer smart system
The Optimizer Smart system has undergone several clinical studies, including randomised controlled studies, in which it improved patients’ six-minute hall walk distance and everyday life quality.
The functional status of the New York Heart Association (NYHA) Class III patients with symptomatic heart failure despite taking medical treatment was also improved by delivering CCM therapy using the Optimizer®.
A study demonstrated that 81.5% of patients receiving CCM therapy with the Optimizer experienced improvement in at least one full NYHA functional class.
In May 2023, the company announced the successful completion of the first implantation for the clinical trial INTEGRA-D, which was intended to assess the safety and efficacy of two established cardiac therapies in combination, including CCM therapy delivered by the Optimizer system and an implantable cardioverter defibrillator (ICD) in one device (CCM-D).
The INTEGRA-D study is a multi-centre study of 300 patients from 75 centres. The trial evaluated the combination of CCM with ICD therapy, using the Optimizer® Integra CCM-D system.
The study will evaluate the CCM-D device in treating episodes of ventricular tachycardia or ventricular fibrillation and provide CCM treatment for heart failure. Patients will be monitored for a minimum of two years.
The Optimizer® Integra™ CCM-D system provides CCM therapy to enhance life quality and alleviate heart failure symptoms, along with ICD therapy to address potentially fatal arrhythmias leading to sudden cardiac death.
Continuing research is being conducted to investigate the safety and effectiveness of CCM for individuals experiencing heart failure with a left ventricular ejection fraction ranging from 40% to 60%.