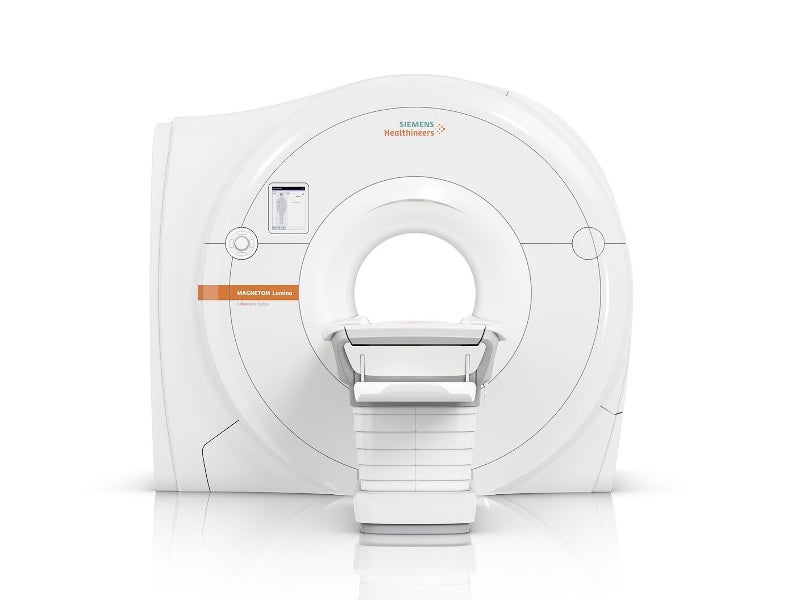
Magnetom Lumina 3 Tesla (3T) magnetic resonance imaging (MRI) system is an Open Bore system developed by Siemens Healthineers.
The device is designed to generate cross-sectional images of the internal structures and functions of the body, including head, body and extremities. It can also be used for producing images during interventional procedures when conducted with MR compatible devices.
The US Food and Drug Administration (FDA) provided 510(k) clearance to the device for human use in January 2019. It was first installed at the Medical University of South Carolina (MUSC) in Charleston, South Carolina in the US in February 2020.
Magnetom Lumina 3 Tesla system technical details
Magnetom Lumina 3T system features a 70cm-wide bore with BioMatrix patient personalisation technology and large 55 x 55 x 50 cm3 field of view (FoV).
Its system measures 1.86m in length, weighs 7.35t and occupies minimum room size of 31m². It has a maximum of 180 channels as well as 32 independent receiver channels configuration, which can be simultaneously used for a single scan and one single FoV, each producing an independent partial image.
The machine comes with XK gradients – 36/200 gradient strength, which delivers powerful neurological, orthopaedics, abdominal and prostate imaging, equipped with ultra-short, lightweight superconducting magnet, which does not require helium refill in normal use for cooling. It is equipped with state-of-the-art zero Helium boil-off technology making the system cost-efficient. Eco-power technology also reduces the energy consumption of the system.
The system consumes 4.3kW when turned off, 8.3kW when ready to measure, while 21.3kW when scanning. The radiofrequency cabin for the system has 90dB damping.
Majority of the system is made of metals and most of the materials used in the device are recyclable. It features actively shielded magnet and gradients with a rechargeable battery with a three-year life cycle.
Details of BioMatrix technology
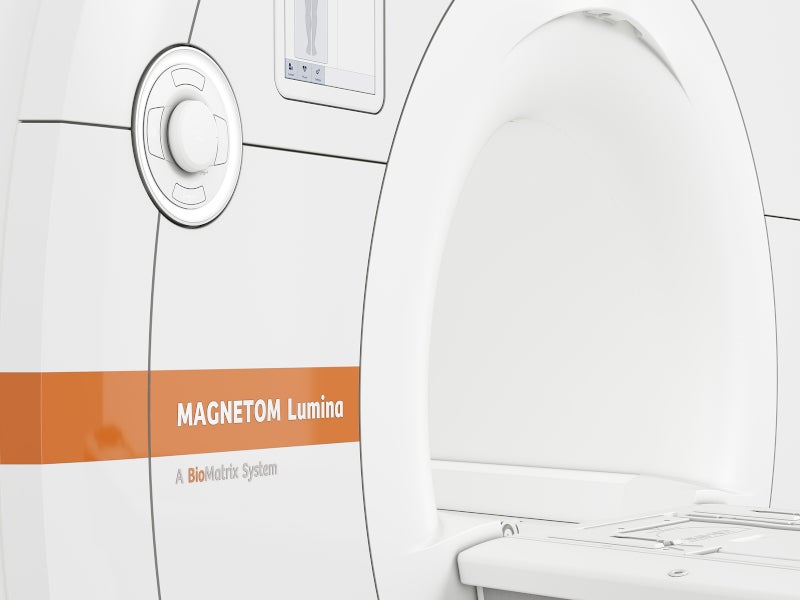
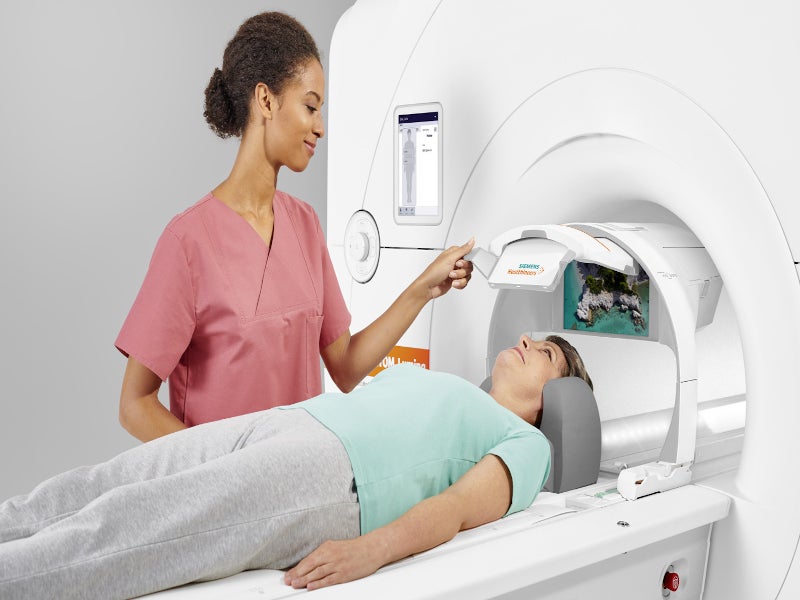
BioMatrix patient personalisation technology of the MRI system features sensors that adapt to the different biological variability of the patients. This enhances productivity while maintaining the quality with the Select&GO Interface, powered by Artificial Intelligence (AI). The technology also accelerates the MRI workflow by permitting rapid patient positioning.
The technology features BioMatrix Tuners, an intelligent coil technology, that adjust to the difficult anatomies of the body. The tuners use CoilShim and SliceAdjust technologies and provide excellent results with homogeneity and fat saturation.
Other technologies and features of Magnetom Lumina 3 Tesla system
Day Optimizing Throughput (Dot) technology of the system brings automation to the system enabling standardised and routine scan procedures highly reproducible. A total of eight Dot engines in the system assist in approximately 90% of all the MRI requests.
The system has syngo Virtual Cockpit to facilitate remote scanning and for delivering standardised and reproducible results across the entire MR system.
MRI system includes the revolutionary Innovision™ infotainment and noise cancellation solution that creates a sound and video experience for patients to reduce anxiety and increase comfort and confidence.
The system is equipped with a wide range of patient-friendly coil portfolio such as Contour 24, a soft and flexible Tim 4G coil design, which adapts to the body shape of the patient. The coil portfolio effectively simplifies and shortens the preparation and scanning duration.
Another feature is the Turbo Suite, which enables customisation of the scanning speed for various areas of the body. This feature nearly halves the scan time without compromising the image quality for routine clinical examinations.
MRI system complies with Environmental Protection, Health Management and Safety (EHS) measures and is certified as per ISO 14001 and OHSAS 18001.