The MAXAIR® Controlled Air Purifying Respirator (CAPR®) system is an advanced respiratory protection equipment approved by the US National Institute for Occupational Safety and Hazard (NIOSH) for protection against aerosolised and airborne particulates.
The personal protective equipment (PPE) meets all US Occupational Safety and Health Administration (OSHA) loose fitting (no Fit Testing required) powered air purifying respirators (PAPR) requirements. It does not require fit testing, which is a significant problem with N95 respirators.
Developed by Bio-Medical Devices International (BMDI), a medical device company based in the US, the system became the respirator of choice for various healthcare organisations amid the 2019 coronavirus outbreak. It provides standard protection for healthcare professionals against Covid-19 in accordance with CDC guidelines
MAXAIR Systems, a part of BMDI, selected Demand Solutions® of Demand Management as its new supply chain planning platform. Demand Management is a wholly-owned subsidiary of US-based supply chain solutions provider Logility. The company implemented Demand Solutions in just three weeks to support the accurate allocation of life-saving inventory in response to the Covid-19 pandemic.
BMDI rapidly increased the manufacture of MAXAIR CAPR in 2020 to keep up with the extreme demand for its highly preferred PAPRs during the Covid-19 pandemic.
MAXAIR CAPR system design and features
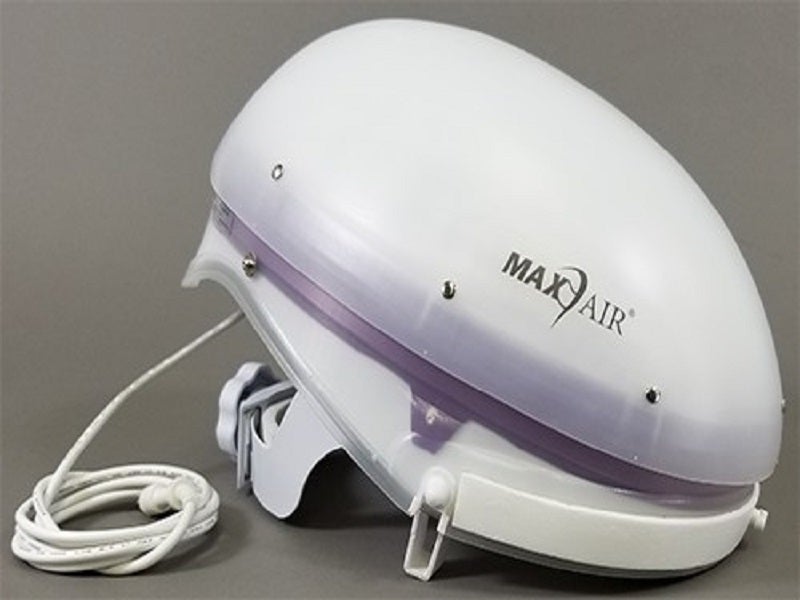
MAXAIR CAPR is a compact, lightweight, controlled computerised, no-hose powered air-purifying respirator (PAPR), whose highly-integrated design avoids the need for large air tubes and heavy belt-mounted blower units present in conventional PAPR.
The system contains four primary components, a helmet with power cord, battery, belt and charger. Various components, including fan, filter, airflow system and motor, are also integrated. The filter cartridge is housed in the helmet, while the filter cover cap (FCC) is securely placed over it. Headband adjustments are positioned for secured wearing.
The airflow switch on the helmet adjusts and operates airflow. The disposable lens cuff (DLC) is placed around the face by fixing it to three attachment points on the helmet. The DLC is a full-face cover with a high assigned protection factor (25).
The LED indicator on the helmet alerts the user of equipment maintenance in real time. The lithium-ion battery pack clips onto the battery belt, which is adjustable and worn around the waist. The system comes with a large and standard battery option.
The 2000-36 standard battery offers a run time of eight to ten hours, while the 2000-37 battery has a longer run time of up to 15 hours. The standard battery takes four to five hours to fully recharge, while the large battery takes five to seven hours. The helmet is connected to the battery by a cord connector.
Shroud and hood configurations with an assigned protection factor (APF) of 1,000 allow full head and neck coverage.
The MAXAIR CAPR System has the HE (High Efficiency Particulate Air) NIOSH Classification, which indicates that it filters at least 99.97% of airborne particles. These HE XP filters can be used every day for eight to 12 months without losing efficiency.
The CAPR System replaces the large hoses and hefty fan units found on existing PAPRs with a thin helmet, which is similar to a bicycle safety helmet.
MAXAIR is the quietest PAPR currently on the market, with normal testing at less than 62dB.
Using the switch on the back of the helmet, the user can adjust the airflow between Low (6 cubic feet per minute (cfm)-7cfm), Medium (7cfm-8cfm) and High (8cfm-9cfm).
The CAPR system is quick and simple to put on and take off, and the risk of self-contamination is reduced owing to an innovative design and fewer parts.
The MAXAIR CAPR system is affordable, as it saves money by eliminating the need to use N95 masks and respirator cartridges.
The simple CAPR system can replace complex and expensive PAPR systems, N95 masks, goggles and basic surgical masks.
MAXAIR CAPR respirator technology
The equipment filters large and small particulates with high efficiency, covering many penetrating particle sizes (MPPS) of 0.04μm to 0.06μm.
This filtration particle size encompasses dust, bacteria, such as Mycobacterium tuberculosis, anthrax and Staphylococcus aureus, and viruses, such as coronavirus, HIV, hepatitis and cytomegalovirus. It combines electrostatic and mechanical filtration technologies to deliver higher filtration efficiency.
Functioning of MAXAIR CAPR system
A fan module pulls air inside the equipment, where it is filtered and then distributed around the face. Positive pressure is maintained in the helmet to facilitate easy breathing without heat, moisture and carbon-dioxide build-up and lens fogging.
The system prevents the inhalation of contaminated air without requiring fit testing, as negative pressure respirators do. It also eliminates facial pressure points.
A blower controlled by a micro-computer facilitates the adjustment of airflow to the user’s desired level and according to their activity level.
Front-mounted LEDs show important data from the airflow microcomputer controller and give the wearer a real-time head-up display (HUD) in the top periphery.
The helmet doesn’t use audio alarms or require manual checks.
Benefits of MAXAIR CAPR
No-hose MAXAIR respirators eliminate the chance of snagging, optimising ease and freedom of movement due to the absence of bulky blower units. System configuration can be conveniently changed into a cuff, shroud or hood.
The system simplifies de-contamination and avoids the need to follow rigorous cleaning procedures. The non-claustrophobic device ensures accurate fit during real-time working conditions to ensure overall comfort and non-interference for healthcare professionals.
The new 2271PB-07 Single and 2272PB-07 Double hood versions provide extra protection against blood-borne pathogens under the ASTM F1671 standard test method.
The configuration allows a full and clear field view for unobstructed communication with patients.